Wavefunctions and Energy Levels
Wavefunctions and Energy Levels. Since particles have wavelike properties cannot expect them to behave like point-like objects moving along precise trajectories.

Wavefunctions and Energy Levels
E N D
Presentation Transcript
Wavefunctions and Energy Levels Since particles have wavelike properties cannot expect them to behave like point-like objects moving along precise trajectories. Erwin Schrödinger: Replace the precise trajectory of particles by a wavefunction (y), a mathematical function that varies with position Max Born: physical interpretation of wavefunctions. Probability of finding a particle in a region is proportional to y2.
y2 is the probability density. To calculate the probability that a particle is in a small region in space multiply y2 by the volume of the region. Probability = y2 (x,y,z) dx dy dz
d2y + V(x) y = E y 2 m dx2 - h Schrödinger Equation The Schrödinger equation describes the motion of a particle of mass m moving in a region where the potential energy is described by V(x). (1-dimension) Only certain wave functions are allowed for the electron in an atom The solutions to the equation defines the wavefunctions and energies of the allowed states An outcome of Schrödinger’s equation is that the particle can only possess certain values of energy, i.e. energy of a particle is quantized.
- h R En = n = 1, 2,... n2 In the H atom the potential that the electron feels is the electrostatic interaction between it and the positive nucleus V(r ) = - e2 / (4 peo r) r: distance between the electron and the nucleus. Solve the Schrödinger equation to determune the allowed energy levels of an electron in the H atom Solution for allowed energy levels is: R = (me e4) / (8 h3eo2) = 3.29 x 1015 Hz
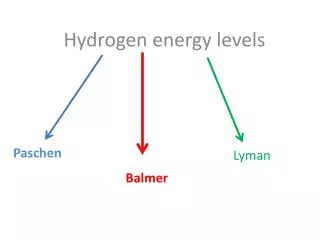
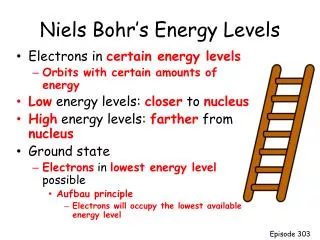
n: principle quantum number. Labels the energy levels When n = 1 => ground state of the H atom. Electron in its lowest energy n > 1 : excited states; energy increases as n increases E = 0 when n = ∞ , electron has left the atom - ionization
Atomic Orbitals Wavefunctions of electrons in atoms are called atomic orbitals, have a dependence on position Square of the wavefunction - probability density of electron The wavefunction of an electron in a hydrogen atom is specified by three quantum numbers, specifying energy and probability of finding an electron. 1) Principle quantum number, n: specifies energy of the orbitals. In a hydrogen atom, all atomic orbitals with the same value of n have the same energy and are said to belong to the same SHELL of the atom.
2) Orbital angular momentum quantum number, l l = 0, 1, 2, …., n-1 Each value of l corresponds to a different type of orbital with a different shape The orbitals of a shell with principal quantum number n fall into n groups, called SUBSHELLS; each subshell is identified by a different l value. l = 0 s-orbitals l = 1 p-orbitals l = 2 d-orbitals l = 3 f-orbitals
Magnetic quantum number, ml : distinguishes the orbitals within a subshell. Determines how the atom behaves in a magnetic field. ml = l, l -1, … - l 2 l + 1 ml values for each l l = 1; ml = +1, 0, -1 n is related to the size of the orbital, l is related to its shape, and ml is related to its orientation in space.
s orbitals: correspond to l = 0 and ml = 0 For Hydrogen atom the ground state is n = 0, l = 0 and ml = 0; a s orbital Density of shading represents the probability of finding an electron at any point. The graph shows how probability varies with distance
Wavefunctions of s orbitals of higher energy have more complicated radial variation with nodes (points of zero probability) Boundary surface encloses surface with a > 90% probability of finding electron
electron density wave function radial probability distribution
f orbitals: Seven f orbitals l = 3, ml = +3, +2, +1, 0 - 1, -2. -3
The three quantum numbers for an electron in a H atom in a certain state are n = 4, l = 2, ml = -1. In what type of orbital is the electron located?
Electron Spin Spectral lines observed did not have exactly the same frequencies as those calculated by Schrödinger. S. Goudsmit and G. Uhlenbeck proposed electrons have spin. Electrons behave like a spinning sphere, like a planet rotating on its axis. An electron has two spin states, represented by and or a and b. Can think of these states as a counterclockwise () spin or a clockwise (), both at the same rate. Spin quantum number, ms, distinguishes the two spin states ms = 1/2 electron ms = - 1/2 electron
The state of an electron in a hydrogen atom is defined by the four quantum numbers, n, l, ml, ms. As the values of n increases, the size of the atom increases.
Many-Electron Atoms Electronic Structure of H atom (Z = 1) Electron in the lowest energy level - ground state of the atom, n = 1 => 1s orbital Quantum numbers of this 1s electron n = 1, l = 0, ml= 0, ms = +1/2 or -1/2 If the electron acquires energy, the electron can undergo a transition to the n = 2 shell and can occupy the 2s or one of the three 2p orbitals (for H-atom all have the same energy) The state of an electron in a H atom is defined by the four quantum numbers n, l, ml, ms. As the value of n increases, the size of the atom increases.
d2y + V(x) y = E y 2 m dx2 - h For H atom: V(r ) = - Z e2 / (4 peo r) (Z = 1 for H atom) Many-electron atoms (Z > 1) Electrons occupy orbitals like those of a H atom. Energies of orbitals of many electron atoms are not the same as those for the H atom. Nuclear attraction for electrons is greater as Z increases lowering the electrons’ energy; also have to account for electron-electron repulsion.
In the Schrödinger equation, V(r ) has to account for both the nuclear-electron attraction and the electron-electron replusion For example for He (Z = 2), V(r ) contains three terms V(r ) = - [(2 e2) / (4 peo r1)] - [(2 e2) / (4 peo r2)] + e2 / (4 peo r12) attraction attraction repulsion For many-electron atoms The electron density of an isolated many-electron atom is ~ sum of the electron densities of each electron taken individually Every electron in an atom has a set of four quantum numbers, n, l, ml and ms
The electron-electron repulsion opposes electron-nuclear attraction. The repulsion “shields” the electron from the full attraction of the nucleus. Electrons feel an “effective” nuclear charge which is less than the full nuclear charge. s orbitals have a non-zero probability density at the nucleus, penetrate through inner shells s electrons feel stronger nuclear attraction; are tightly bound and hence lower in energy p orbitals have zero probability density at the nucleus; less penetrating than s and hence p electrons are higher in energy. d orbitals less penetrating than p and hence d electrons are higher in energy than p
In many electron atoms, because of shielding and penetration effects, order of the energy of orbitals in a given shell is typically s < p < d < f. Energies of orbitals depend on both n and l (not just n as in the H atom)
Exclusion Principle The electronic structure of an atom determines its chemical properties. Electron configuration - a list of all occupied orbitals of an atom, with the number of electrons that each contains Pauli Exclusion Principle: No more than two electrons may occupy any given orbital. When two electrons occupy an orbital their spins must be paired. No two electrons in an atom can have the same set of quantum numbers.
(a) Spins are paired if one is and the other . Paired spins denoted as ; ms of each is different (b) Two electrons have parallel spin if both spins are in the same direction
Building Up: fill orbitals starting with the lowest energy (aufbau principle), pairing electrons as determined by the Pauli principle. Order of energies of orbitals 5f 6d 7p 8s 4f 5d 6p 7s 3f 4d 5p 6s 3d 4p 5s 3p 4s 2p 3s 2s 1s