Enhancing Forensic Sexual Assault Casework with Laser Microdissection (LMD) Techniques
Laser Microdissection (LMD) presents a novel approach to streamline the processing of forensic sexual assault samples. In the North Louisiana Criminalistics Laboratory, LMD helps eliminate manual extraction bottlenecks, ensures reproducibility and sensitivity, and allows for direct amplification of DNA from samples. This technique effectively separates sperm and epithelial DNA, reducing analysis time and improving accuracy. Although initial costs may be high, the benefits of enhanced efficiency and minimized PCR interference make LMD a valuable tool in forensic science.

Enhancing Forensic Sexual Assault Casework with Laser Microdissection (LMD) Techniques
E N D
Presentation Transcript
Application of Laser Microdissection (LMD) to Expedite Forensic Sexual Assault Casework Kelli Raley, MSFS North Louisiana Criminalistics Laboratory Shreveport, LA NFSTC Laser Microdissection Workshop, 2007
LMD Microscopy Research and Experience: Highlights • Why LMD for North Louisiana Crime Lab (NLCL) Casework? • Elimination of Manual Extraction • Reproducibility/Sensitivity • Troubleshooting • Single Amplification/Optimization • Absence of Sperm • LMD: Streamlined, Novel Process
Why Laser Microdissection? ~45% of all NLCL DNA cases involve sexual offense so Need to eliminate bottleneck in DNA analysis
Disadvantages of LMD • Initial cost • Novel validation • ~$4.25 each for PEN slides • PEN foil contains pores ≈ sperm • Except for PEN slides, no other consumables for Leica
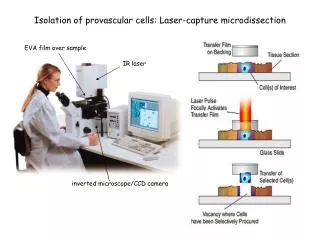
Advantages of LMD • Eliminate traditional extraction • Absolute separation of sperm and epithelial DNA • Effect of traditional PCR challenges minimized • Decrease analysis time for a sexual assault sample
Elimination of Traditional Extraction • Direct amplification after LMD?
Elimination of Traditional Extraction • Direct amplification after LMD? • Pre-amplification Lysis • Constraints • Can’t adversely affect PCR • Limited volume (10µL) • Lyse-N-Go™ PCR Reagent (LNG) • 25µL reaction vs. 50µL • Recombinant Proteinase K
Comparison of Pre-Amplification Treatments • 150 sperm • 25µL volume • Profiler Plus • 30 PCR cycles
Pre-amplification Lysis: ProK/DTT • Cut directly into water • Recombinant ProK • Lysis incubation in TC • PCR reaction components to same tube • Amplify, analyze
Additional Experiments: Sensitivity • PCR cycles • amplification reaction volume with reduced volume PCR (RVPCR) • 15µL, 10µL • PCR with fewer sperm for lowest detection limit
25µL 15µL 10µL ProK/DTT150 sperm, 30 cycles Avg PH ~880 RFUs Avg PH ~948 RFUs Avg PH ~2540 RFUs
Reproducibility • Profiler Plus • 30 PCR cycles
drop-out ProK/DTT100 sperm, 30 cycles 25µL Avg PH ~157 RFUs 10µL Avg PH ~2631 RFUs
drop-out ProK/DTT50 sperm, 30 cycles 25µL Avg PH ~174 RFUs 10µL Avg PH ~556 RFUs
LMD together with Pre-amplification Lysis: • Elimination of traditional extraction possible • Absolute separation of sperm and epithelial DNA possible
Exciting! 50 sperm, 30 cycles, 10µL Profiler Plus
drop-out 50 epi nuclei, 30 cycles, 10µL Profiler Plus
drop-out 25 epi nuclei, 30 cycles, 10µL Profiler Plus
Early NLCL Research for LMD: Summary • Replace DNA extraction and purification with novel pre-amplification lysis • Physical and complete separation of sperm and epithelial DNA • RVPCR, 30 cycles, reproducible • Sensitivity: 50-150 sperm • http://www.promega.com/geneticidproc/ussymp16proc/abstracts/langley.pdf
From Research to Validation . . . • Troubleshooting • Identifiler for single amplification • How many sperm? • No sperm observed
Troubleshooting • Static • humidity • cuttings into 25µL vs 50µL • Electropherogram Conundrum • “dye-saturated” e-grams, • Contamination? • Non-specific binding? • Profiler Plus vs. Profiler vs. Identifiler
From Research to Validation . . . • Troubleshooting • Identifiler for single amplification • How many sperm? • No sperm observed
Identifiler Optimization • TE-4 vs DepC H2O • Vary Tris, pH 8.0 in TE-4: normal, 1/2x, 1/4x, 1/5x • PCR cycling • 28+6, 20+14, 20+10 cycles • 31 cycles • MgCl2 • Vary extra Mg2+ added • 0.5mM – 1.5mM still optimal for RVPCR
50 sperm, Identifiler, 30 cycles drop out 24/29
Identifiler Optimization 29/29 alleles
From Research to Validation . . . • Troubleshooting • Identifiler for single amplification • How many sperm? • No sperm observed
50 sperm, Identifiler 29/29 alleles
25 sperm, Identifiler 23/29
25 sperm, Identifiler 27/29
15 sperm, Identifiler 20/29
15 sperm, Identifiler 25/29
From Research to Validation . . . • Troubleshooting • Identifiler for single amplification • How many sperm? • No sperm observed
No sperm observed • Cut spot from PEN slide • Organic extraction • Qiagen EZ1? • YSTR and STR panels
Advantages of LMD • Eliminate traditional extraction • Absolute separation of sperm and epithelial DNA • Effect of traditional PCR challenges minimized • Decrease analysis time for a sexual assault sample through novel process
Advantages of LMD Effect of traditional PCR challenges minimized • Simplify mixtures, simplifying interpretation • Difficult statistical interpretations eliminated • Less tendency for contaminants/inhibitors? • Increase PCR cycles to enhance LCN sperm analysis
Novel Process The NLCL DNA section envisions a new way of processing and storing sexual assault samples
Sexual Assault Sample Processing Time - LMD • Presumptive testing (AP, PSA), slide preparation • Examine for sperm, cut nuclear material • Drying of TE-4 • Pre-amp (in TC) • PCR and gel 2 hours (1hr shake) 15 min -1 hour 45 min overnight (3.5 hrs) 1 day
LMD Coupled With Pre-amplification Lysis . . . . . Improves and streamlines the analysis of sexual assault evidence AND Frees up analyst time!
Current Considerations • Shorten lysis time of LMD harvested cells • Mixture Studies • Epithelial nuclei, still necessary? • Non-probative samples • Considerations for casework implementation