Matter :
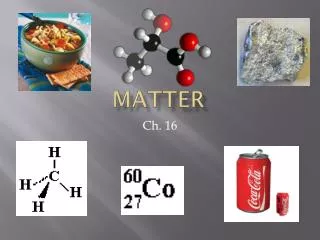
Matter :. States and Properties. What is matter?. Matter is anything that has mass and volume (takes up space.). What is a physical property?. A physical property is something that can be observed or measured without changing the matter’s identity .

Matter :
E N D
Presentation Transcript
Matter: States and Properties
What is matter? • Matter is anything that has mass and volume (takes up space.)
What is a physical property? • A physical property is something that can be observed or measured without changing the matter’s identity. Some physical properties of matter include: mass, weight, volume, density, color, and odor.
What is mass? What is volume? • Mass is the amount of matter in an object. • Volume is the amount of space a object takes up.
What is weight? • Weight is the pull of gravityon the massof an object.
What is density? • Density is the amount of mass in a given space or volume. • D= M/V • What has more density: a pound of feathers or a pound of lead? or
Why do liquids of different densities form layers? • The densest layer is also the heaviest…it settles on the bottom. Other layers appear in the order of most dense to least dense. Molasses
What are the three main states of matter? • A state is a physical form. • Matter is commonly either a solid, a liquid or a gas, the three common states of matter.
What is a solid? • Solids have definite shape and volume. • Two types of solids are: • Amorphous: • its particles are not arranged in a pattern. • Crystalline: • its particles have an orderly repeating pattern
Amorphous form Solids
Crystalline form Solids
What is a liquid? • State of matter that changes shape but not volume. • Liquids take the shape of their container.
What is the container in these photos of a liquid? Water
What is a gas? • State of matter that changes both shape and volume. • Particles spread out as far as container will allow.
What is plasma? • Plasma is superheated gas • Plasma can create electrical and magnetic fields.
Plasma Sun with solar flares (plasma)
How does one state of matter become another? • Matter changes state because of changes in temperature and pressure
Ways Matter Changes State: • Melting • Melting is the change from a solid to a liquid state • Takes place at the melting point • Particles move faster • Endothermic reaction
Ways Matter Changes State: • Freezing • Freezing is the change from a liquid to a solid state • Takes place at the freezing point • Particles slow down • Energy removed • Exothermic reaction
Ways Matter Changes State • Evaporation • Evaporation is the change from liquid to gas Evaporation happens at the surface of a liquid *not the same as boiling, which occurs all through the liquid
Ways Matter Changes State • Boiling • Boiling is the change from liquid to gas • Boiling takes place throughout the liquid • Occurs when the vapor pressure equals the atmospheric pressure • boiling and melting points are the same for any substance • Changes with elevation
Ways Matter Changes State • Condensation • Condensation occurs when a gas changes state to a liquid • The condensation point is the temperature and pressure that the gas changes to a liquid. • Same temperature as boiling point • Condensation is the reverse of evaporation
Ways Matter Changes State • Sublimation • Sublimation is the change from solid directly to gas • Endothermic reaction where energy is gained as it changes state • Examples: dry ice and snow packs can sublimate without going through the liquid state
What is a chemical property? • A chemical property is any characteristic that gives a substance the ability to undergo a change that results in a new substance. • Example: A match burns and becomes ash.
What is a chemical property? • Chemical properties describe what chemical changes can occur • Flammability: the ability to combust or burn • Reactivity: the ability to combine with other substances
What is a chemical change? • Chemical change: • When substances change into new substances that have different properties.
Examples of chemical changes • Sour milk
Other Examples of Chemical Changes • Include the: combustion (burning) of wood, metabolism of food in the body, mixing an acid and a base such as hydrochloric acid (HCl) and sodium hydroxide (NaOH), cooking an egg, digesting sugar with the amylase in saliva, mixing baking soda and vinegar to produce carbon dioxide gas, baking a cake, electroplating a metal or using a chemical battery!
How do you know if a chemical change has occurred? Signs of a chemical change: 1. Change in odor 2. Heat is released or cooling occurs 3. Sound or light 4. Color 5. Fizzing and foaming
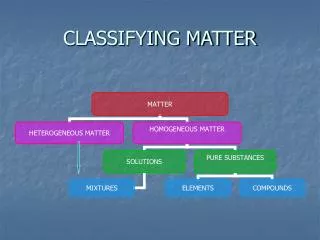
What is a mixture? • A mixture is a combination of two or more substances that are not chemically combined • Examples: Cowboy pizza (pepperoni, sausage and Canadian beacon) Chorizo Omelette Peanut Butter Sandwich
How do you separate a mixture? • Distillation • Separates based on different boiling points • Evaporation • Removal of water • Filtration • Screening out larger particles • Physical Methods • Pick out the different substances
What is a compound? • A compound is a pure substance made up of two or more elements that have been chemically combined
What is the difference between Mixtures and Compounds? • Compounds: • Elements combine chemically • Elements become new substance with different properties • Elements cannot be separated • Mixtures: • elements mixed, not chemically combined • Elements keep their individual properties • Elements can be separated
What is a solution? • A solution is a mixture that appears to be a single substance • Particles are evenly distributed among each other • Solutions may be liquids, gasses or solids
What is an atom? • An atom is the smallest particle into which an element can be divided and still be the same substance. • Atoms are composed of even smaller particles
Parts of the Atom: Protons are in the nucleus (center) of the atom. Protons have a positive charge Proton have much of the mass in an atom
Parts of the Atom: Neutrons are also in the nucleus of the atom. Neutrons have no charge Neutrons are also about as massive as protons
Parts of the Atom: Electrons are outside the nucleus of the atom are. Electrons have a negative charge Electrons have very small mass, only about 1/1800 of a proton.
What are the properties of elements? • Elements are made up of a single type of atom. • Their properties include: • Density, or mass/volume • Melting Point, the temperature solid changes to liquid • Conductivity, the ability to carry electricity • Hardness, or how resistant to scratching a substance is • Color, determined by what wavelengths of light are absorbed
How are elements classified? Metals: • Found to the left of the zigzag line on periodic table • Most elements are metals • Most are solid at room temperature • Have few electrons in outer levels
How are elements classified? • Nonmetals: • Found to the right of zigzag line • Many are gasses at room temperature • Have nearly complete set of electrons in outer levels.
How are elements classified? • Metalloids: • Found on border of the zigzag line on the periodic table • Have about half the electrons in outer levels. • Have some properties of both metals and nonmetals