Structure and Function of the Kidney
410 likes | 2.42k Vues
Structure and Function of the Kidney. The functional unit of the kidney is the nephron The major functions of the kidney are to maintain extracellular fluids, to eliminate wastes resulting from normal metabolism, and to excrete xenobiotics and their metabolites

Structure and Function of the Kidney
E N D
Presentation Transcript
Structure and Function of the Kidney • The functional unit of the kidney is the nephron • The major functions of the kidney are to maintain extracellular fluids, to eliminate wastes resulting from normal metabolism, and to excrete xenobiotics and their metabolites • Mammalian kidneys have 10,000-1,000,000 nephrons per kidney
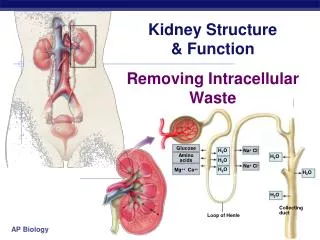
Structure and Function of the Kidney (cont) • The glomerulus yields an ultrafiltrate of plasma that represents 20% of the renal blood flow, ie. 2-3% of cardiac output • Endothelial surface is negatively charged and contains fenestrae • The glomerular basement membrane is sandwiched between the epithelial cells and contains anionic sialoglycoproteins, glycoproteins and collagen IV • The mesangium provides support • The outer capsule is Bowman’s capsule
Structure and Function of the Kidney (cont) • The tubule resorbs greater than 99% of the glomerular filtrate • The proximal tubule has extensive resorption and selective secretion (convoluted - S1 and S2, straight - S3). S2 is primary site for low MW protein resorption and S3 is primary site for P450. • Thin loop of Henle - resorption of fluids • Distal tubule - resorption of fluids and acid-base balance • Collecting duct - resorption of fluids, antidiuretic hormone and acid-base balance
Structure and Function of the Kidney (cont) • Produces erythropoietin, which regulates RBC production • Hydroxylates 25-OH-cholecalciferol (vitamin D metabolite), to promote bone resorption and calcium and phosphorus absorption from the gut • Releases renin to regulate the peripheral renin-angiotensin-aldosterone system (juctaglomerular apparatus)
Assessment of Kidney Function:Morphologic Evaluation • Urinalysis • Gross evaluation of the kidney at necropsy • Histopathology of the kidney • Electron microscopy of the kidney
Assessment of Kidney Function:Urinalysis • Proteinuria - indicates glomerular damage • Glycosuria - indicates tubular damage • Urine volume and osmolarity • pH • Enzymes - indicates tubular damage • Microscopic examination - casts, crystals, bacteria, etc.
Assessment of Kidney Function:Blood Chemistries • Blood urea nitrogen (BUN) • Creatinine • Electrolytes - Ca, Mg, K, P • Glomerular filtration rate - determines the clearance of inulin, creatinine and BUN • Renal clearance - measures the clearance of p-aminohippuric acid by filtration and secretion
Glomerular Disease: Toxicities due to Alteration of Anionic Charge • Hexadimethrine - polycationic molecule reduces anionic charge, which permits escape of anionic molecules such as albumin and IgG • Polynucleoside of puromycin - damages epithelial foot processes
Glomerular Disease:Immune Complex Disease • Anti-GBM mediated glomerulonephritis is induced by heterologous antibodies • Antibodies due to exogenous antigens - cationized molecules such as lysozyme, IgG and BSA bind to anionized surfaces; Concanavalin A binds to sugars in the GBM
Glomerular Disease:Immune Complex Disease (cont) Deposition of circulating immune complexes • Drug or toxin-induced T-cell dependent polyclonal B-cell activation - mercury in Brown Norway rats • Unknown mechanism - gold salts, D-penicillamine, hydralazine • Antibodies to heterologous proteins - safety evaluations of recombinant proteins in laboratory animals
Nephrosis: Damage to the renal tubule • Halogenated hydrocarbons - chloroform, hexachlorobutadiene, trichloroethylene, dibromochloropropane, & bromobenzene • Heavy metals - cadmium, mercury & lead • Antibiotics - cephalosporins & aminoglycosides • Mycotoxins - ochratoxin A & citrinin • Ethylene glycol • Antineoplastic drugs - cisplatinum • Alpha2u-globulin nephropathy
Haloalkane Nephrosis • Chloroform is metabolized by P450 to an electrophile, phosgene, which is a potent cytotoxicant. • Carbon tetrachloride is metabolized to free radicals and phosgene. • P450 is localized in the proximal tubule. • This results in nephrosis with necrosis, enzyme, glucose and protein excretion in urine, and increased BUN and creatinine concentrations in serum.
Haloalkene Nephrotoxicity • 1,1-Dichloroethylene, trichloroethylene and tetrachloroethylene are metabolized by P450 to electrophilic metabolites and or free radicals. • These metabolites can be cytotoxic and/or genotoxic. • Nephrotoxicity is exacerbated when glutathione is depleted.
Glutathione-mediated Nephrosis • Glutathione conjugates of haloalkanes can form episulfonium ions. • Primary route for 1,2-dichloroethane, 1,2-dibromoethane and 1,2-dibromo-3-chloro-propane. • These can alkylate macromolecules and cause cytotoxicity and genotoxicity.
Cystine Conjugate -lyase Activation • Stable cystine conjugates from glutathione can be formed in the liver from trichloro-ethylene, tetrafluoroethylene and hexa-chlorobutadiene and transported to the kidney. • They are further metabolized by -lyase in the kidney to generate reactive thiols.
Biotransformation of Trichloroethylene + GSH GSH-transferase Cytochrome P450 -Lyase Chlorothioketene
Trichloroethylene-associated renal cell carcinomas • Highly exposed workers exhibit nephrosis and an increased incidence of renal cancer • TCE and other renal neoplasms have a high incidence of mutations in the von Hipple-Lindau (VHL) tumor suppressor gene. This requires mutation of one allele and deletion of the other • TCE mutations include a hot spot at nucleotide 454 (CT) • VHL gene is located at 3p25 • 3p25.5 is a fragile site at the chromosome telomere • ogg1 mutations are also associated with renal cancer and it is also located at 3p25
Lead Nephropathy • Lead induces acute nephrosis of the proximal tubule and affects blood pressure to the kidney. • Low molecular weight proteins bind Pb2+, leading to resorption by endocytosis. • Cellular and mitochondrial swelling, karyomegaly, mitosis and intranuclear inculsion bodies are seen. • Decreased tubular resorption of glucose, phosphate and amino acids +/- proteinuria • Chronic exposure causes tubulo-interstitial disease.
Cadmium Nephrotoxicity • Metalothionein (Mt) is a low molecular weight protein that transports cadmium (Cd) to the kidney. • Mt is taken up in the lysosomes and Cd is released. Cd t ½ is 10-30 years in humans. • Free Cd stimulates synthesis of renal Mt, which binds Cd. When renal Mt is depleted, toxicity occurs in S1 and S2. • Proteinuria, calciuria, aminoaciduria and glucosuria are seen. • Chronic exposure leads to tubulo-interstitial disease. There appears to be a threshold of 200 ppm Cd in the kidney before renal disease occurs.
Mercury Nephropathy • Hg2+ and methylmercury cause damage to the S3 and S2/S3 segments of the proximal tubule. • Hg2+ causes damage to the cell membrane and mitochondria, and loss of cellular control over intracellular Ca++. • Chronic exposure results in anti-GBM and immune-complex disease of the kidney. • Methylmercury is highly lipid soluble and concentrates in the proximal tubule, causing damage to mitochondria and lysosomes.
Chromium Nephropathy • Cr+6 causes acute nephrosis of S1 and S2. • It damages the brush border, causing decreased absorption of proteins, glucose and amino acids. • Large membranous myeloid bodies form as a result of disrupted lysosomal function. • Decreased glomerular filtration leads to increased BUN and creatinine.
Ethylene Glycol Nephropathy • Toxicity first discovered in humans when ethylene glycol was used as a drug solvent. • Causes acute renal toxicity in animals after exposure to anti-freeze. • Ethylene glycol is metabolized to oxalic acid. • Calcium oxylate crystals form in the lumen of the tubules.
Renal Papillary Necrosis • Caused by non-steroidal anti-inflammatory drugs – phenacetin and acetaminophen. • Highest concentration of drugs are in the renal papilla. • Prostaglandin hydroperoxidase is highest in the medulla and is thought to metabolize these drugs to reactive quinoneimines.
Urinary Bladder Toxins • Chemicals that cause bladder stones and calcium phosphate precipitates frequently cause bladder cancer in rodents. • Aromatic amines, tobacco smoke and parasitic infections cause bladder cancer in humans.
Saccharin • Non-genotoxic • Induces bladder epithelial tumors in male rats at >1% in the diet • Must be administered to neonatal animals • Can act as a promoter for genotoxic agents • Only the sodium salt of saccharin is carcinogenic • Mechanism requires the formation of calcium phosphate-containing precipitates