Chapter 3 Structures and Functions of Nucleic Acids
1.26k likes | 3.16k Vues
Chapter 3 Structures and Functions of Nucleic Acids. Nucleic acid. A biopolymer composed of nucleotides linked in a linear sequential order through 3’,5’ phosphodiester bonds. Classification of nucleic acid. Ribonucleic acid (RNA) is composed of ribonucleotides .

Chapter 3 Structures and Functions of Nucleic Acids
E N D
Presentation Transcript
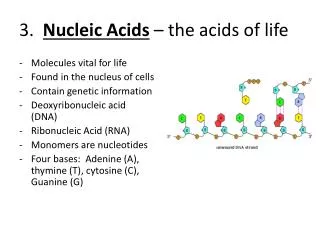
Nucleic acid A biopolymer composed of nucleotides linked in a linear sequential order through 3’,5’ phosphodiester bonds
Classification of nucleic acid • Ribonucleic acid (RNA) is composed of ribonucleotides. • in nucleiand cytoplasm • participate in the gene expression • Deoxyribonucleic acid (DNA)is composed of deoxyribonucleotides. • 90% in nuclei and the rest in mitochondria • store and carry genetic information; determine the genotype of cells
Interesting history • 1944: proved DNA is genetic materials(Avery et al.) • 1953: discovered DNA double helix (Watson and Crick) • 1968: decoded the genetic codes (Nirenberg) • 1975: discovered reverse transcriptase (Temin and Baltimore) • 1981: invented DNA sequencing method (Gilbert and Sanger) • 1985: invented PCR technique(Mullis) • 1987: launched the human genome project • 1994: HGP in China • 2001: accomplished the draft map of human genome
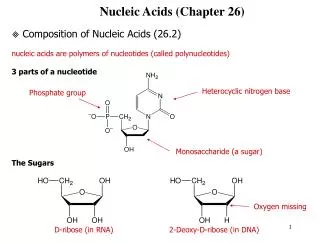
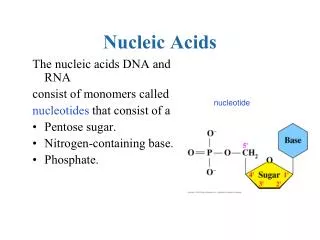
§ 1.1 Molecular Constituents Nucleic acid can be hydrolyzed into nucleotides by nucleases. The hydrolyzed nucleic acid has equal quantity of base, pentose and phosphate. phosphate pentose nucleic acid nucleotides nucleosides bases
Adenine (A) Guanine (G) Base: Purine
Uracil (U) Thymine (T) Cytosine (C) Base: Pyrimidine
5´ 1´ 4´ 3´ 2´ -D-ribose -D-2-deoxyribose Pentose
1 1´ Ribonucleoside glycosidic bond Purine N-9 or pyrimidine N-1 is connected to pentose (or deoxypentose) C-1’ through a glycosidic bond.
Ribonucleotide phosphoester bond A nucleoside (or deoxynucleoside) and a phosphoric acid are linked together through the 5’-phosphoester bond.
AMP ADP ATP Nucleic acid derivatives Multiple phosphate nucleotides • adenosine monophosphate (AMP) • adenosine diphosphate (ADP) • adenosine triphosphate (ATP)
cAMP Nucleic acid derivatives Cyclic ribonucleotide: 3’,5’-cAMP, 3’,5’-cGMP, used in signal transduction
Nucleic acid derivatives Biologically active systems containing ribonucleotide: NAD+, NADP+, CoA-SH
Phosphoester bond formation The -P atom of the triphosphate group of a dNTP attacks the C-3’ OH group of a nucleotide or an existing DNA chain, and forms a 3’-phosphoester bond.
Nucleic acid chain extension A nucleic acid chain, having a phosphate group at 5’ end and a -OH group at 3’ end, can only be extended from the 3’ end.
Phosphodiester bonds Alternative phosphodiester bonds and pentoses constitute the 5’-3’ backbone of nucleic acids.
§ 2.1 Primary Structure • The primary structure of DNA and RNA is defined as the nucleotidesequence in the 5’ – 3’ direction. • Since the difference among nucleotides is the bases, the primary structure of DNA and RNA is actually the base sequence. • The nucleotide chain can be as long as thousands and even more, so that the base sequence variations create phenomenal genetic information.
§ 2.2 Secondary structure The secondary structure is defined as the relative spatial position of all the atoms of nucleotide residues.
§ 2.2.a Chargaff’s rules • The base composition of DNA generally varies from one species to another. • DNA isolated from different tissues of the same species have the same base composition. • The base composition of DNA in a given species does not change with its age, nutritional state, and environmental variations. • The molarity of A equals to that of T, and the molarity of G is equal to that of C.
A G C T A/T G/C G+C Pu/Py E. coli 26.0 24.9 25.2 23.9 1.09 0.99 50.1 1.04 Tuberculosis 15.1 34.9 35.4 14.6 1.03 0.99 70.3 1.00 Yeast 31.7 18.3 17.4 32.6 0.97 1.05 35.7 1.00 Cow 29.0 21.2 21.2 28.7 1.01 1.00 42.4 1.01 Pig 29.8 20.7 20.7 29.1 1.02 1.00 41.4 1.01 Human 30.4 19.9 19.9 30.1 1.01 1.00 39.8 1.01 Molarity of bases
Building a milestone of life James Watson and Francis Crick proposed a double helix model of DNA in 1953. It symbolized the new era of modern biology.
§ 2.2.b Double helix of DNA • Two DNA strands coil together around the same axis to form a right-handed double helix (also called duplex). • The two strands run in opposite directions, i.e., antiparallel. • There are 10 base pairs or 3.4nm per turn and the diameter of the helix is 2.0nm.
Backbone and bases The hydrophilic backbone is on the outside of the duplex, and the bases lie in the inner portion of the duplex.
Base interactions • The two strands of DNA are stabilized by the base interactions. • The bases on one strand are paired with the complementary bases on another strand through H-bonds, namely G≡C and A=T. • The paired bases are nearly planarand perpendicular to helical axis. • Two adjacent base pairs have base-stacking interactions to further enhance the stability of the duplex.
Groove binding Small molecules like drugs bind in the minor groove, whereas particular protein motifs can interact with the major grooves.
§ 2.2.c Polymorphisms of DNA • DNA can resume different forms depending upon their chemical microenvironment, such as ionic strength and relative humidity. • B-form DNA is the predominant structure in the aqueous environment of the cells. • A-form and Z-form are also native structures found in biological systems.
Hoogsteen base pair The third strand is using Hoogsteen H-bonds to pair with bases on the first strand.
G-quartet DNA • The telomere of DNA is a G-righ sequence, such as • 5’ (TTGGGG)n 3’ • 4 G residues constitute a plane which is stabilized by Hoogsteen H-bonds.
G-quartet of DNA Four strands are arranged in either parallel or antiparallel manner.
§ 2.3 Supercoil Structure §2.3.a Supercoil structure • The two termini of a linear DNA could be joined to form a circular DNA. • The circular DNA is supercoiled, and supercoil can be either positive or negative. • Only the supercoiled DNA demonstrate biological activities.
EM image of supercoiled DNA Circular DNAs in nature, in general, are negatively supercoiled.
§2.3.b Prokaryotic DNA • Most prokaryotic DNAs are supercoiled. • Different regions have different degrees of supercoiled structures. • About 200 nts will have a supercoil on average.
§2.3.c Eukaryotic DNA • DNA in eukaryotic cells ishighly packed. • DNA appears in a highly ordered form called chromosomes during metaphase, whereas shows a relatively loose form of chromatin in other phases. • The basic unit ofchromatin is nucleosome. • Nucleosomes are composed of DNA and histone proteins.