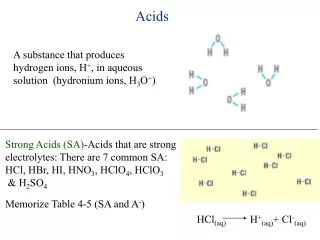
Acids
Acids. and Salts. Bases. What do all these acids have in common:. Weak Acid The Formula. Acetic acid (vinegar) HC 2 H 3 O 2 Carbonic acid HCO 3. Acid:. A substance that ionises (breaks down into ions) and releases H + ions in an aqueous solution. “aqueous” means: water.


Acids
E N D
Presentation Transcript
Acids and Salts Bases
What do all these acids have in common: Weak AcidThe Formula Acetic acid (vinegar) HC2H3O2 Carbonic acid HCO3
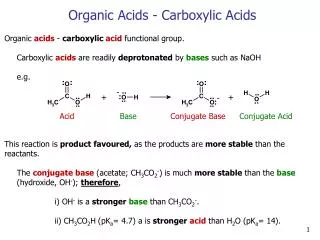
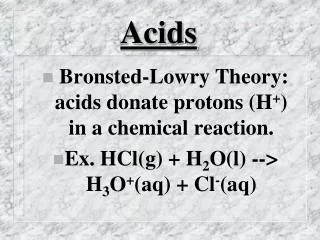
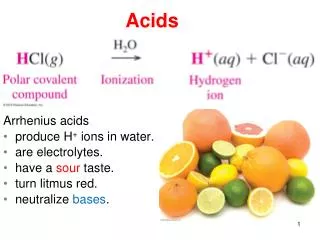
Acid: A substance that ionises (breaks down into ions) and releases H+ ions in an aqueous solution “aqueous” means: water
Characteristicsof Acids: • Acids have a sour taste • Acids react with metals (to produce hydrogen gas) • Acids have a pH of 1-6 • Acids turn universal indicator yellow, orange or red • Acids turn litmus red • Many are corrosive or poisonous
You need to know three acids • Sulfuric acid • H2SO4 • hydrochloric acid • HCl • Nitric acid • HNO3
All are Strong Acids Ionise (break down into ions) completely to give off many H+ ions
Weak Acids (only partially breaks down (ionise), gives less H+)
Base: A substance that ionises and releases OH- ions in an aqueous solution Alkali: A base that dissolves in water
Characteristics of Bases: • Bases usually taste bitter • Bases feel slippery • Bases have a pH of 8 – 14 • Bases turn universal indicator blue or purple • Bases turn litmus blue OH-
BUT AREN’T METAL OXIDES BASES? • Yes!!!!!! • Metal oxide + water metal hydroxide • Metal oxides release hydroxide ions in solution
Some bases (VERY FEW) don’t have or produce OH- Examples include: ammonia NH3 Carbonates (CO3 and HCO3) ***Any chemical which neutralizes an acid can be considered a base
Indicators An indicator is a compound that will change color in the presence of an acid or base • Red Litmus-Turns blue in base • Blue Litmus-Turns red in acid • Universal indicator (pH paper) Used for the full pH range • Phenolphthalein-Turns pink in base
pH pH stands for “potential of Hydrogen” and is a measure of how many H+ ions there are in solution. The MORE H+ there are, the LOWER the pH will be.
pH Scale Shows the range of H+ concentrations High H+ concentration Low H+ concentration
Reactions between acids and bases When an acid and a base react with each other , the characteristic properties of both are destroyed. This is called neutralisation. This happens because equal amounts of the two substances cancel each others properties and produce two neutral substances (water and a salt).
Reactions between acids and bases General formula for acid base reaction: → + Water + Base Salt Acid “Salt” means any ionic compound formed from an acid/base reaction NOT JUST NaCl !!
Neutralisation HCl + NaOH → H2O + NaCl acid base water salt
Neutralisation Another Example HCl + KOH → H2O + KCl H Cl K OH acid base water salt
Complete and incomplete neutralisation • Any time an acid and base react neutralisation will occur. However, whether the solutions are fully neutralised depends on several factors: • The pH of both reactants • The concentration of both reactants • The quantity of both reactants • If any of these factors occur then neutralisation will be incomplete. This means in the solution there will be a metal salt, water and an acid or base.
Name the following compounds • CaO • NaOH • Na2S • PbCO3 • CuHCO3 • Ag2O • PbCl2 • CuSO4
General equations • These are all extensions of the acid + base equations • Acid + metal oxide metal salt + water • Acid + metal hydroxide Metal salt + water • Acid + Metal carbonate metal salt+ water + carbon dioxide • Acid + metal hydrogen carbonate metal salt + water+ carbon dioxide
WHAT SALT IS FORMED DEPENDS ON WHAT ACID IS USED • Hydrochloric acid makes a chloride salt • Sulfuric acid makes a sulfate salt • Nitric acid makes a nitrate salt
Predict what acid these salts were made from • NaNO3 • CaSO4 • PbCl2 • CuCl2 • Fe2(SO4)3 • Fe(NO3)2
Complete the following as word equations only • Hydrochloric acid + Calcium hydroxide • Sulphuric acid + Lead oxide • Nitric acid + sodium bi-carbonate • Silver oxide + sulphuric acid • Copper carbonate + nitric acid
2. Balancing word equations • Calcium oxide + Hydrochloric acid Calcium chloride + water • Nitric acid + copper carbonate copper nitrate + water + carbon dioxide • Hydrochloric acid + lead hydroxide lead chloride + water • Copper oxide + sulphuric acid copper sulphate + water • Sodium hydrogen carbonate + nitric acid sodium nitrate + water + carbon dioxide
NCEA 2009 • A student carried out an experiment to neutralise sulfuric acid by adding sodium hydroxide to it. • Discuss how the student could have determined when the sulfuric acid had been neutralised and what effect adding the sodium hydroxide has on the pH of the solution. • In your answer include: • • an explanation of neutralisation in terms of an acid-base reaction • • the name of the indicator used • • observations that the student would make as the sodium hydroxide is added to the acid • • a word and balanced chemical equation for the reaction.
NCEA 2006 • The pH values of three substances are given below: • Hydrochloric acid pH = 1 • Potassium carbonate solution pH = 9 • Sodium hydroxide solution pH = 14 • (a) When Universal Indicator solution is added to each of these substances, what colour would result? • (i) Hydrochloric acid __________________ • (ii) Potassium carbonate solution __________________ • (iii) Sodium hydroxide solution __________________ • Potassium carbonate solution is added slowly to the hydrochloric acid (without indicator) in a beaker until no further change is seen. • (b) (i) Describe what you would see happening when potassium carbonate is added to the acid. • (ii) Write a balanced chemical equation for the reaction between potassium carbonate and hydrochloric acid.
NCEA QUESTION • HCl is added to a solution of sodium hydroxide. The reaction takes place but complete neutralisation did not occur. Discuss three things that could have lead to this.
TRY THESE – IN WORDS AND THEN BALANCED CHEMICAL EQUATIONS • Hydrochloric acid + potassium hydroxide • Sulfuric acid + calcium oxide • Hydrochloric acid + potassium hydroxide • Sulfuric acid + sodium hydroxide