Animal Cell Bioreactor
470 likes | 937 Vues
Animal Cell Bioreactor. Introduction . Animal cell bioreactor depends on cell itself whether: The cell exhibit anchorage- dependent growth characteristics that it must be attached to some form of solid matrix support. Cells can grow and produce when in suspended culture.

Animal Cell Bioreactor
E N D
Presentation Transcript
Introduction • Animal cell bioreactor depends on cell itself whether: • The cell exhibit anchorage- dependent growth characteristics that it must be attached to some form of solid matrix support. • Cells can grow and produce when in suspended culture. • Cells are completely surface-dependent. • Animal cell bioreactor deals with cells that are larger, more fragile and more complex than bacteria. The cells are more subject to damage by osmotic, shear and other cytotoxic effects. • Furthermore, cell productivities are low and the cultures more liable to contamination by faster growing bacteria or fungal cells.
Bioreactor design for animal cell should consider : • Surface adherence properties of the cells • Increased demand on asceptic procedures • The shear sensitivity of the cells • Potential foaming in the medium • Potential mass transfer limitations • Control and automation • Ease of scale up • Removal of toxic waste products
Animal Cell Bioreactor Operation Batch, Fed-batch, Repeated Fed-batch,Continuous, and Perfusion system.
Batch • Condition in the medium change continuously with respect to time, owing to the depletion of substrates and the build-up of the waste products. • Provides the cells with a highly fluctuating and uncontrolled environment which can therefore never be optimal. Fed-batch and Repeated fed-batch Advantages • Supply of nutrients to the cells can be controlled to some extent by the feeding policy, and also have special application in processes where products are obtained during the stationary stages of growth.
Continuos Operation (Chemostat & Turbidostat) Advantages • More constant and controlled condition. • Steady state operation at controlled specific growth rate or cell density conditions. • Specific nutrient limitations may be easily effected • Deliberate manipulation of the cell physiology for optimal productivity can be obtained. • Reduce capital cost, increase media utilization, reduced downtime and removal of waste products of reactor environment is continuously. • However, it requires good control facilities and high standards of plant containment to avoid contamination.
Perfusion system. • Cells provided with a continuous supply of fresh medium and continuous withdrawal of waste products. • Closely approach the in vivo system and appear particularly to be advantages in cases in which the kinetics of product formation are related to growth. • Often used in conjunction with an external loop, which is used to recirculate medium to the cells at controlled temp, dissolved O2, pH and etc. • Fine mesh rotating metal filters mounted on the agitator shaft, give high cell retention rates and good scale-up potential for continuous operation.
Figure 11.11(a) semi-batch reactor with control of pH, Po2 and rate of medium additional.
Fig 11.11(b) perfused chemostat operation with cell retention filter.
Animal cell bioreactor design Attached Growth Systems
Attached growth systems • PROBLEMS OF SCALE-UP: -need to maintain a uniform cell & medium distribution -hard to control cell environment -hard to monitor growth processes (difficult to do direct measurement of cell concentration) • Types of bioreactor considered based on: a) surface anchorage cells, b) cell density values, c) surface area/medium volume ratios employed • Microcarrier, immobilized bed, and plate heat exchanger reactors are really efficient respect to high scale up potential • Surface dependent cells required:- large surface area to volume ratio- enough nutrient supply- removal of waste- ease of harvesting
Continue… 5) Stacked Plate Unit • Plates maybe used in a static or rotational mode • Poorly it need very large quantity of medium volume per unit surface area (V/A)
Animal cell bioreactor design Suspended Growth Systems
Advantage of suspension system: • Easy to scale up. • rate of cell growth is easy to monitor. • the environment condition is easy to obtained.
Traditional method of suspension culture-spinner flask. • Used for low volumes (200ml-20L). • The conventional stirred tank bioreactor can also be used for small to large scales of operation.
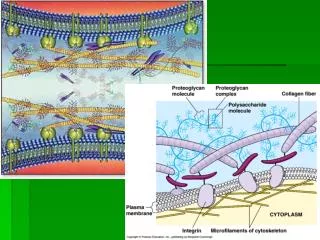
Characteristics needed in animal cell bioreactors: • Combination of round-bottomed vessels with slow moving propeller agitators are preferred. • Vibromix agitators may be used for uniform power dissipation but are unsuitable for microcarrier applications. • Fermenter wall surfaces must be smooth • The vessels have no baffles, to minimize shear.
In stirred tank bioreactor operating at low densities, sufficient oxygen transfer for dissolved oxygen was control by regulating the oxygen partial pressure in the bioreactor using a nitrogen and air gas mixture. • As growth proceeds, this may be suplemented by additional oxygen by the direct sparging. The head space usually contain CO2 as a means of regulating pH. • This means that control of dissolved oxygen and pH are strongly important and can lead to contradictory requirements. • Increase in supply of CO2 at high pH value=lower partial pressure of O2 and lower oxygen supply rate
Airlift bioreactor are attractive for animal cell culture: • Reduce the contamination-absence of any moving parts or mechanical seals. • Shear conditions is low • Possibility to control dissolved oxygen and CO2 by varying the rate and composition of the gas flow through the bioreactor.
Animal cell bioreactor design Immobilized cell systems
Immobilization Technique • An immobilized enzyme is an enzyme which is attached to an inert, insoluble material such as sodium alginate. This can provide increased resistance to changes in conditions such as pH or temperature. It also allows enzymes to be held in place throughout the reaction, following which they are easily separated from the products and may be used again • It is well known that pure enzymes change their behavior, their stability, when they are immobilized. In the past two decades the immobilization of microorganisms, cells and parts of cells was gradually introduced into microbiology and biotechnology. • The immobilized whole cell system is an alternative to enzyme immobilization. Unlike enzyme immobilization, where the enzyme is attached to a substrate (such as calcium alginate), in immobilized whole cell systems, the target cell is immobilized
According to A.H. Scragg (1988) many of the cell immobilization techniques are modifications of the techniques developed for enzymes. However, the larger size of microbes has influenced the techniques • In industrial , methods for cell immobilization are categories under two broad categories that is : i) immobilized-“free” cell method ii) modified “cell” method
In the free-cell method, cells are immobilized by confining them behind dialysis/ filtration membranes. Examples are hollow fibre, membrane sheet reactor, ceramic matrix, encapsulated and entrapped cell system. • In modified cell system, examples are show in table 1
Immobilized cell reactor develop for • To provide systems of high cell density • Continual harvesting of product can be obtained at high productivity when combined high throughput perfusion system • Design to minimize problems of nutrient and waste product diffusion.
Cells grow in the extracapillary space of a hollow cartridge but with transfer of oxygen • removal of waste products occurring via the combination of a cell exclusion membrane • product exclusion or ultrafiltration membrane with a cut-off chosen to exclude, but passing low-molecular weight metabolites. • The cell growth is perfused directly by medium. • A highly oxygenated medium stream on the other side of this membrane provides sufficient oxygen transfer. • PH control is effected by a secondary loop of medium on the product exclusion membrane side.
Similar principle was proposed but comprising stacked membranes with support. • Alternate microfiltration and ultrafiltration membranes provide a system of a separate chamber, which contain medium, cells and product respectively. • The cells are immobilized in a wire net matrix between the differing membrane sheets, from the medium chamber, are fed by nutrient and oxygen diffusing via the ultrafiltration membraneHigh molecular- weight products similarly diffuse via the microfiltration membrane into the product number
The factor need to consider when using cell immobilization technique: • Choice of matrix material • Special sensitivity of the cells to environmental shock • Natural material such as gelatine, alginate, hydrogel, and agrose are generally preferred.
Crossflow consumables To remove mammalian cells from liquid.