Boron
E N D
Presentation Transcript
Discovery of the Element • Sir Humphrey Davy, Gay-Lussac and Thenard discovered boron in 1808 • Compounds of boron have been known of for thousands of years. • In early Egypt, mummification depended upon an ore known as natron, which contained borates as well as some other common salts. • Borax glazes were used in China from 300 AD, and boron compounds were used in glassmaking in ancient Rome. • The first pure boron was produced by the American chemist W. Weintraub in 1909, although this is disputed by some researchers. • Turkey and the U.S. are the world's largest producers of boron.
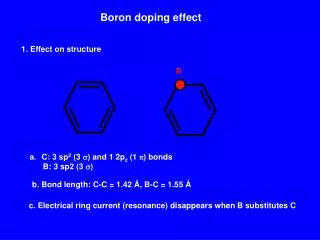
Characteristics of the Element • Boron contains boron atoms randomly bonded to each other without long range order. • Crystalline boron, a very hard material with a high melting point, exists in many polymorphs. • Optical characteristics of crystalline/metallic boron include the transmittance of infrared light. • At standard temperatures, metallic boron is a poor electrical conductor, but is a good electrical conductor at high temperatures. • Chemically boron is electron-deficient, possessing a vacant p-orbital. It is an electrophile. • Compounds of boron often behave as Lewis acids, readily bonding with electron-rich substances to compensate for boron's electron deficiency. • The reactions of boron are dominated by such requirement for electrons. • boron is the least electronegative non-metal, meaning that it is usually oxidized (loses electrons) in reactions. • Boron is also similar to carbon with its capability to form stable covalently bonded molecular networks.
Source of the Element • Much boron is found in chemical compounds in its ore borax. • Boron is never found free in nature. • Two types of boron are found (allotropes). • Amorphous boron is a brown powder and metallic (crystalline) boron is black and hard and a weak conductor at room temperature. • Pure boron is used as a dopant (a substance added to semiconductors to change how it behaves with electricity) in the semiconductor industry. • Chemical compounds of boron are important as to make strong materials not weigh very much, as nontoxic insecticides and preservatives, and for chemical synthesis. • Plants need Boron in them to live • Very small amounts of boron are needed in animal's bodies so that they are very healthy. • Important sources of boron are ore rasorite (kernite) and tincal (borax ore). • Both of these ores are found in the Mojave Desert -Tincal is the most important source of boron from the Mojave. • Extensive borax deposits are also found in Turkey. • Boron of 99.9999% purity has been produced and is available commercially.
Uses of the Element • amorphous boron is used in pyrotechnic flares (distinctive green colour), and rockets (as an igniter) • boric, or boracic, acid, is used as a mild antiseptic • borax, Na2B4O7.10H2O, is a cleansing flux in welding • borax, Na2B4O7.10H2O is a water softener in washing powders • boron compounds are used in production of enamels for covering steel of refrigerators, washing machines, etc. • boron compounds are extensively used in the manufacture of enamels and borosilicate glasses • boron compounds show promise in treating arthritis • 10B is used as a control for nuclear reactors, as a shield for nuclear radiation, and in instruments used for detecting neutrons • boron nitride is as hard as diamond. It behaves like an electrical insulator, but conducts heat like a metal. It also has lubricating properties similar to graphite • the hydrides are sometimes used as rocket fuels • boron filaments, a high-strength, lightweight material, are used for advanced aerospace structures, . • lightweight compounds used for aerospace structures • boron filaments used in fibre optics research • Boric Acid is also used in North America for the control of cockroaches, silverfish, ants, fleas, and other insects.