The Typical Cell
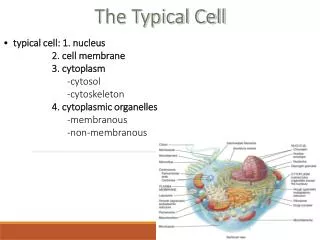
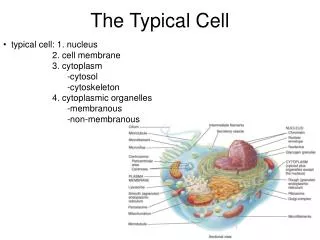
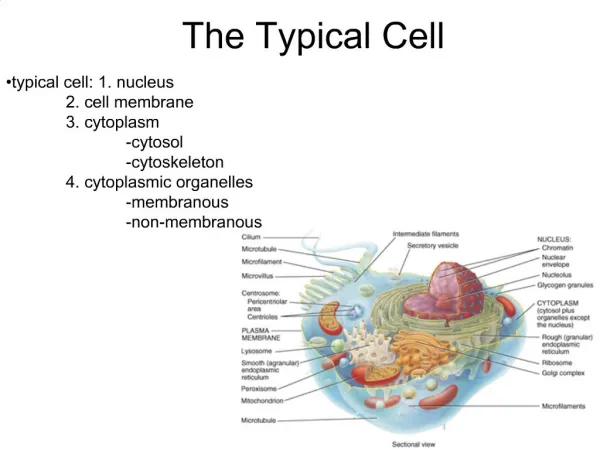
The Typical Cell. typical cell: 1. nucleus 2. cell membrane 3. cytoplasm -cytosol -cytoskeleton 4. cytoplasmic organelles -membranous -non-membranous. Cytoplasm. semi-fluid-like jelly within the cell division into three subdivisions:

The Typical Cell
E N D
Presentation Transcript
The Typical Cell • typical cell: 1. nucleus • 2. cell membrane • 3. cytoplasm • -cytosol • -cytoskeleton • 4. cytoplasmic organelles • -membranous • -non-membranous
Cytoplasm • semi-fluid-like jelly within the cell • division into three subdivisions: • 1. cytosol • 2. supportive framework of proteins: cytoskeleton • 3. organelles – membranous and non-membranous
Cytosol ECF • lower K+ • higher Na+ • lower concentration • of dissolved and • suspended proteins • higher concentration • of carbohydrates • smaller reserves of amino • acids • higher K+ • lower Na+ • higher concentration • of dissolved and • suspended proteins • (enzymes, organelles) • lower concentration • of carbohydrates • (due to catabolism) • larger reserves of amino • acids (anabolism) The Cytosol – Eukaryotic Cells • eukaryotic cells – part of the cytoplasm • about 55% of the cell’s volume • about 70-90% water PLUS • ions • dissolved nutrients – e.g. glucose • soluble and insoluble proteins • waste products • macromolecules and their components - amino acids, fatty acids • ATP • unique composition with respect to extracellular fluids
Cytoskeleton: • internal framework of the cell • gives the cytoplasm flexibility and strength • provides the cell with mechanical support • gives the cell its shape • anchorage points for organelles and cytoplasmic enzymes • also plays a role in cell migration and movement by the cell
Cytoskeleton: • three major components • 1. microfilaments • 2. intermediate filaments • 3. microtubules
5 m 10 m 10 m Column of tubulin dimers Keratin proteins Actin subunit Fibrous subunit (keratinscoiled together) 25 nm 812 nm 7 nm Tubulin dimer
1. microfilaments = thin filaments made up of a protein called actin -solid rods of about 7nm -twisted double chain of actin subunits -forms a dense network immediately under the PM (called the cortex) -also found scattered throughout the cytoplasm
microfilaments = -function: 1.anchor integral proteins and attaches them to the cytoplasm 2. interaction with myosin = interacts with larger microfilaments made up of myosin - results in active movements within a cell (e.g. muscle cell contraction) 3. provide much of the mechanical strength of the cell – resists pulling forces within the cell 4. give the cell its shape 5. also provide support for cellular extensions called microvilli (small intestines)
2. intermediate filaments = more permanent part of the cytoskeleton than other filaments • - range from 8 to 12 nm in diameter • five types of IF filaments – type I to type V • made up of proteins such as vimentin, desmin, or keratin • each cell type has a unique complement of IFs in their cytoskeleton • all cells have lamin IFs – but these are found in the nucleus • some cells also have specific IFs • e.g neurons also posses IFs made of neurofilaments type I IFs = acidic keratins type II IFs = basic keratins type III IFs = desmin, vimentin type IV IFs = neurofilaments type V IFs = nuclear lamins kidney cell - vimentin
2. intermediate filaments = function: 1. impart strength to the cytoskeleton – specialized for bearing tension (like microfilaments) 2. support cell shape e.g. forms the axons of neurons 3. anchors & stabilize organelles e.g. anchors the nucleus in place 4. transport materials e.g. movement of neurotrasmitters into the axon terminals
3. microtubules= hollow rods or “straws” of 25 nm in diameter • - made of repeating units of proteins called tubulin • tubulin is a dimer – two slightly different protein subunits • called alpha and beta-tubulin -the basic microtubule is a hollow cylinder = 13 rows made up of tubulin b-tubulin a-tubulin
3. microtubules - function: 1. cell shape & strength 2. organelles: anchorage & movement 3. mitosis - form the spindle(chromosome movement) 4. form many of the non-membranous organelles - cilia, flagella, centrioles b-tubulin a-tubulin
can be found as a single tube a doublet and a triplet 3. microtubules -animal cells – microtubule assembly occurs in the MTOC(microtubule organizing center or centrosome) -area of protein located near the nucleus -within the MTOC : 1. assembling MTs 2. a pair of modified MTs - called centrioles -other eukaryotes – there is no MTOC -have other centers for MT assembly
Non-membranous Organelles A. Centrioles:short cylinders of tubulin - 9 microtubule triplets -called a 9+0 array (9 peripheral triplets, 0 in the center) -grouped together as pairs – arranged perpendicular to one another -make up part of the centrosome or MTOC -possible role in MT assembly -also has rolein mitosis - spindle and chromosome alignment
B. Cilia & Flagella • cilia = projections off of the plasma membrane of eukaryotic cells – covered with PM BUT NOT MEMBRANOUS ORGANELLES • about 0.25um in diameter and only 20um long • beat rhythmically to transport material – power & recovery strokes • found in linings of several major organs covered with mucus where they function in cleaning e.g. trachea, lungs Trachea
B. Cilia & Flagella 0.1 m Plasma membrane Outer microtubuledoublet Dynein proteins • cytoskeletal framework of a cilia or flagella = axoneme(built of microtubules) • contain 9 groups of microtubule doublets surrounding a central pair= called a 9+2 array • cilia is anchored to a basal body just beneath the cell surface Centralmicrotubule Radialspoke Microtubules Cross-linkingproteins betweenouter doublets (b) Cross section ofmotile cilium Plasmamembrane Basal body 0.1 m 0.5 m (a) Longitudinal sectionof motile cilium Triplet (c) Cross section ofbasal body
flagella = resemble cilia • -much larger – 9+2 array • -found singly per cell • -functions to move a cell through the ECF • -DO NOT HAVE THE SAME STRUCTURE AS BACTERIAL FLAGELLA
Membranous Organelles • completely surrounded by a phospholipid bilayer similar to the PM • surrounding the cell • allows for isolation of each individual organelle - so that the interior of each organelle does not mix with the cytosol • -known as compartmentalization • BUT - cellular compartments must “talk” to each other • therefore the cell requires a well-coordinated transport system in order for the organelles to communicate and function together • -”vesicular transport”
Membranous Organelles • major functions of the organelles • 1. protein synthesis & secretory pathway – ER and Golgi • 2. energy production – mitochondria • 3. waste management – lysosomes and peroxisomes
Overview of the secretory pathway • Proteins that traffic through the ER-Golgi have three destinations: • Outside the cell = secreted proteins • Plasma membrane • Lysosome = lysosomal enzymes
Endoplasmic reticulum (ER) = series of membrane-bound, flattened sacs in communication with the nucleus and the PM • -ER is typically more than half of the total membrane of an average animal cell • -each sac or layer = cisternae • -inside or each sac =lumen (10% of total cell volume) • -distinct regions of the ER are functionally specialized – Rough vs. Smooth ER
Endoplasmic reticulum (ER) = -three functions: 1. synthesis – lipids and proteins – Rough ER 2. storage – intracellular calcium – Smooth ER 3. transport – site of transport vesicle production – Smooth ER
Rough Endoplasmic reticulum (ER) -outside studded with ribosomes -continuous with the nuclear membrane -protein synthesis, phospholipid synthesis -initial site of processing and sorting of proteins for transport to the Golgi • two kinds of proteins enter the RER: • ER proteins • 2. proteins destined for the Golgi, plasma membrane, lysosome or secretion
Modifications in the RER • 1. formation of disulfide bonds • 2. folding of the peptide chain • 3. addition and processing of carbohydrates • 4. breakage of specific peptide bonds – proteolytic cleavage • 5. assembly into multimeric proteins (more than one chain) • for an animation go to http://sumanasinc.com/webcontent/animations/content/proteinsecretion_mb.html
Import of Proteins into the RER • transport across the ER membrane requires the presence of an ERsignal sequence (red in the figure) • a complex of proteins will bind this signal in the cytoplasm = signal recognition particle/SRP • the ER membrane has receptor for the SRP and ribosome – SRP receptor (yellow protein in figure) • the ribosome is docked next to a “hole” in the ER membrane (blue protein in figure) = translocon • the translocon recognizes the signal in the polypeptide and binds it – “guides” the rest of the translating polypeptide into the ER lumen • once the polypeptide is fed into the ER lumen – a peptidase (located in the SRP receptor complex) cleaves the signal sequence off http://www.rockefeller.edu/pubinfo/proteintarget.html
Smooth ER– extends from the RER • free of ribosomes • many enzymes found on the surface of the SER • 1. lipid synthesis for membranes • 2. steroid biosynthesis(e.g. estrogen and testosterone) • 3. detoxificationof toxins and drugs • 4. transport vesicle formation • 5. cleaves glucose so it can be released into the bloodstream
2. Ribosomes = can be considered a nonmembranous organelle • 2 protein subunits in combination with RNA • -large subunit = ribosomal RNA + ~50 proteins • -small subunit = rRNA + ~33 proteins • found in association with the ER = where the peptide strand is fed into from the ribosome • also float freely within the cytoplasm as groups = polyribosomes
3. Golgi Apparatus = stack of 3 to 20 flattened membrane sacs/cisternae • movement of proteins from the RER - through the stacks via transport vesicles • -definite direction: first stack = cis-face (from the RER, protein modification) • middle stack =medial-face (adds carbohydrates) • last stack = trans-face (modification and packaging into vesicles)
3. Golgi Apparatus • site of final protein modification and packaging of the finished protein • functions: • 1. major site of carbohydrate addition to proteins = glycosylation • 2. site for phosphate addition to proteins = phosphorylation • 3. production of sugars • 4. formation of the lysosome • 5. packaging of proteins and transport to their final destination • Golgi acts as a sorting station for transport vesicles
Modifications in the Golgi = Glycosylation • glycosylation = produces a glycoprotein or a proteoglycan • most plasma membrane and secreted proteins have one or more carbohydrate chains that help target them to the correct location • O-linked sugars are added one at a time in the Golgi (usually 4 subunits total) • N-linked sugars are added as a group (about 14 sugars!) in the ER Proteoglycan
Modifications in the Golgi • glycosylation starts in the ER • N-linked glycosylation – addition of N-linked oligosaccharides • for the proper folding of the protein • glycosylation continues in the Golgi • addition of O-linked oligosaccharides to proteins • the sugars are found in the cytoplasm but are transported into the ER & Golgi by specific transporters for their addition by transferase enzymes
Why Glycosylation? • O-linked glycosylation • O-linked are added one at a time in the Golgi to the amino acids serine, threonine or lysine (one to four saccharide subunits total) • added on by enzymes called glycosyltransferases • human A, B and O antigens are sugars added onto proteins and lipids in the plasma membrane of the RBC • everyone has the glycosyltransferase needed to produce the O antigen • those with blood type A have an additional Golgi glycosyltransferase enzyme which modifies the O antigen to make the A antigen • a different glycosyltransferase is required to make the B antigen • both glycosyltransferases are required for the creation of the AB antigen • coded for by specific gene alleles on chromosome 9 (ABO locus)
Modifications in the Golgi: Protein Trimming • some PM proteins and most secretory proteins are synthesized as larger, inactive pro-proteins that will require additional processing to become active • e.g. albumin, insulin, glucagon • this processing occurs very late in maturation • processing is catalyzed by protein-specific enzymes called proteases • some proteases are unique to the specific secretory protein • occurs in secretory vesicles that bud from the trans-Golgi face • processing could be at one site (albumin) others may require more than one peptide bond (insulin)
The Golgi: Protein transport within the cytoplasm • protein transport within the cell is tightly regulated • most proteins usually contain “tag” or signals that tell them where to go • in the Golgi - specific sequences within a protein will cause: • 1. retention in the Golgi • 2. will target it to lysosomes • 3. send it to the PM for fusion • 4. send it to the PM for secretion • a lack of a signal means you will automatically be secreted = constitutive secretion
SO - WHERE DO PROTEINS GO AFTER THE GOLGI??? -proteins budding off the Golgi have three targets: • targets: • secretory vesicles for exocytosis • membrane vesicles for incorporation into PM • transport vesicles for the lysosome • e.g. digestive enzymes
WHAT IF YOU AREN’T ONE OF THESE PROTEINS?? • ER proteins stay in the ER • never traffic to the Golgi • these ER proteins will have a retention signal – unique group of amino acids (KDEL) • Ribosomal proteins • translation of ribosomal proteins are done in the cytoplasm by polyribosomes • assembled into the large and small protein subunits in the cytoplasm • imported into the nucleus • 28S and 18SrRNA’s are transcribed in the nucleolus - no translation • protein subunits and rRNA’s are assembled in the nucleolus to form the 40S and 60S ribosomal subunits – exported from nucleus • mitochondrial proteins • the mitochondria has its own DNA, transcribes its own mRNA and has its own ribosomes for translation
4. Mitochondria = site of energy production (ATP production) -via Cellular Respiration - breakdown of glucose results in the production of ATP -initial glucose breakdown occurs in the cytosol = Glycolysis -terminal stages occur in the mitochondria = Oxidative Phosphorylation -has its own DNA - maternal -reproduce themselves via dividing
4. Mitochondria • -surrounded by a dual phospholipid bilayer • an outer mitochondrial membrane • an inner mitochondrial membrane • a fluid-filled space = mitochondrial matrix (contains ribosomes!) • -the inner membrane is folded into • folds calledcristae • -these increase the membrane surface area for the enzymes of Oxidative Phosphorylation
outermembrane - 50% phospholipid & 50% protein • -very permeable - contains pores for the import and export of critical materials • innermembrane - 20% phospholipid & 80% protein • -less permeable vs. the outer membrane • -folded extensively to form partitions = cristae • -contains proteins that work to create an electrochemical gradient • -contains enzymes that use this gradient for the synthesis of ATP • -also contains pumps to move ATP into the cytosol • matrix - lumen of the mitochondria • -breakdown of glucose into water and CO2 ends here (enzymes of the Transition phase Kreb’s Cycle)
Cellular Respiration -glycolysis -transition phase -citric acid cycle -electron transport chain http://biology.about.com/gi/dynamic/offsite.htm?site=http://www.sp.uconn.edu/%7Eterry/images/anim/ETS.html http://biology.about.com/gi/dynamic/offsite.htm?site=http://www.biocarta.com/pathfiles/krebPathway.asp http://vcell.ndsu.nodak.edu/animations/etc/movie.htm
Glycolysis phosphorylation (ATP used) isomerization phosphorylation (ATP used) 2 ATP consumed no energy created • literally means “splitting sugar” • conversion of glucose (6 carbon sugar) into 2 molecules of pyruvate (3 carbon sugar) • results in the production of 2 ATP and 2 NADH molecules • reactions of glycolysis take place in the cytosol cleavage 4 ATP created 2 ATP net 2 NADH created http://web.indstate.edu/thcme/mwking/glycolysis.html http://science.nhmccd.edu/biol/glylysis/glylysis.html
Glycolysis phosphorylation (ATP used) isomerization phosphorylation (ATP used) 2 ATP consumed no energy created • under aerobic conditions - pyruvate is converted into acetyl-coenzyme A(Acetyl-CoA) which then enters the citric acid cycle/Kreb’s cycle • under anaerobic conditions glucose is oxidized into pyruvate then converted into lactate (lactic acid) cleavage 4 ATP created 2 ATP net 2 NADH created http://web.indstate.edu/thcme/mwking/glycolysis.html http://science.nhmccd.edu/biol/glylysis/glylysis.html
1 2 3 Transition Phase: Oxidation of Pyruvate to Acetyl CoA • before the citric acid cycle can begin- pyruvate must be converted to acetyl Coenzyme A (acetyl CoA) • links glycolysis to the citric acid cycle • pyruvate is pumped into the matrix of the mitochondria by a transport protein • converted into acetyl coA by a multimeric protein complex called pyruvate dehydrogense complex • forms the waste product carbon dioxide (2 molecules) MITOCHONDRION CYTOSOL CO2 Coenzyme A Acetyl CoA + H NADH NAD Pyruvate • results in the creation of 2 more NADH Transport protein
Pyruvate processing • all kinds of organisms convert glucose to pyruvate using similar reactions • however pyruvate can be processed in many ways to produce • 1. acetyl CoA– the majority of pyruvate is processed by the transition phase • 2. lactate– by cells in the absence of oxygen • 3. ethanol– by yeast • pyruvate acetylaldehyde ethanol • regulated by aldehyde dehydrogenase • a DH = removes H+ from one substrate and adds it to another
Pyruvate processing: Transition Phase • ethanol aldehyde • catalyzed by alcohol dehydrogenase (ADH) • ADH is expressed in humans by the gastric epithelium • there are 5 different alleles for making ADH • ADH2 and ADH3 genes show polymorphism – different forms in different populations • unprocessed alcohol has multiple targets • The overall effect is to slow the functional processes of the brain cell. GABA is commonly known as the brain's "brake" mechanism. • toxic effects = drunk
Citric Acid cycle • named after Hans Adolf Krebs • Acetyl-CoA is converted into oxaloacetic acid -> citric acid • the citric acid is converted into a series of compounds that eventually regenerates OA acid • Cycle “turns” twice – one for each pyruvate • while this cycle only runs in the presence of oxygen – no oxygen is used
Citric Acid cycle • Cycle results in the formation of NAD+ (nicotinamide adenine dinucleotide) • compound capable of storing high energy electrons • as the cycle runs NAD+ is reduced to form NADH and H+ (gains two electrons) • NADH = electron carrier • this NADH will then enter the electron transport chain • Cycle also produces another electron carrier FADH2 • 2 “turns” results in the creation of 6 NADH, 2 FADH2 and 4 CO2
Electron transport chain • electrons are transferred to oxygen eventually forming water • electrons are transferred by a series of protein complexes in the inner mitochondrial membrane • these complexes also serve as proton pumps – pump protons (taken from water) into the space between the two mitochondrial membranes cytochrome c oxidase
cytochrome c reductase cytochrome c oxidase NADH dehydrogenase Electron transport chain • NADH transfers 2 electrons to NADH dehydrogenase • the electrons are then moved from NADH dehydrogenase to cytochrome c reductase (by Q = ubiquinone) and then to cytochrome c oxidase (by cytochrome c) • each of these enzyme complexes pump protons into the intermembrane space - to creates a proton gradient= source of potential energy • as protons flow down their gradient back into the matrix, they pass through an enzyme complex called ATP synthase– which synthesizes ATP protons electrons
ETC animations • http://www.youtube.com/watch?v=xbJ0nbzt5Kw&feature=relmfu • http://www.youtube.com/watch?v=3y1dO4nNaKY • http://biology.about.com/gi/dynamic/offsite.htm?site=http://www.sp.uconn.edu/%7Eterry/images/anim/ETS.html