Name Reactions B.Sc.II P-V By N. M.Gosavi
Name Reactions B.Sc.II P-V By N. M.Gosavi. . Synthesis and Reactions of β- Dicarbonyl compounds. Introduction Compounds having two carbonyl groups separated by an intervening carbon are called β-dicarbonyl compunds, and these compounds are highly versatile reagents for organic synthesis.


Name Reactions B.Sc.II P-V By N. M.Gosavi
E N D
Presentation Transcript
Name Reactions B.Sc.II P-V By N. M.Gosavi
. Synthesis and Reactions of β- Dicarbonyl compounds Introduction Compounds having two carbonyl groups separated by an intervening carbon are called β-dicarbonyl compunds, and these compounds are highly versatile reagents for organic synthesis.
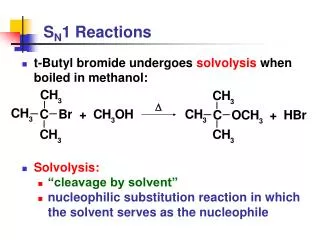
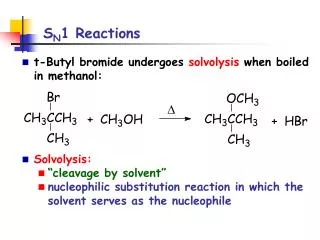
The claisen condensation: The synthesis of β-keto esters When ethyl acetate reacts with sodium ethoxide, it undergoes a condensation reaction. After acidification, the product is a β-keto esters , Condensations of this type occur with many other esters and are known generally as claisen condensation (克莱森缩合)
Crossed Claisen condensations Crossed Claisen condensations are possible when one ester component has no α-hydrogens and is, therefore, unable to undergo self-condensation.
The knoevenagel condensation Active hydrogen compounds condense with aldehydes and ketones known as Knoevenagel condensations, these aldol-like condensations are catalyzed by weak bases. An example is the following:
The Mannich reaction Active hydrogen compounds react with formaldehyde and a primary or secondary amine to yield compounds called Mannich bases.
Cannizaro’s Reaction:- Statement :- Aldehydes which do not contain α hydrogen atom undergoes self oxidation and reduction to produce corrosponding alcohol and carboxylic acid. Generally Benzaldehyde and formaldehyde are used.
The Cannizzaro reaction uses an aldehyde with no hydrogens on the -carbon and NaOH. Since there are no -hydrogens to remove with the base an aldol reaction can not occur. • Hydroxide ion attacks one of the carbonyls forming a tetrahedral intermediate. Reforming the pi bond results in transfer of a hydride (H:) to an unreacted carbonyl in another molecule of the aldehyde.