The Flow of Energy— Part one
260 likes | 291 Vues
Explore the flow of energy and temperature changes in various processes, including exothermic and endothermic reactions. Learn about thermochemistry, heat transfer mechanisms, and the laws of thermodynamics.

The Flow of Energy— Part one
E N D
Presentation Transcript
The Flow of Energy— Part one 17.1 • The temperature of lava from a volcano ranges from 550°C to 1400°C. As lava flows, it loses heat and begins to cool. You will learn about heat flow and why some substances cool down or heat up more quickly than others.
17.1 Energy Transformations • Energy Transformations • In what direction does heat flow?
17.1 Energy Transformations • Heat, represented by q, is energy that transfers from one object to another because of a temperature difference between them. • Heat always flows from a warmer object to a cooler object.
17.1 Energy Transformations • Thermochemistry is the study of energy changes that occur during chemical reactions and changes in state. • The energy stored in the chemical bonds of a substance is called chemical potential energy.
17.1 Energy Transformations • When fuel is burned in a car engine, chemical potential energy is released and is used to do work.
17.1 Exothermic and Endothermic Processes • Exothermic and Endothermic Processes • What happens in endothermic and exothermic processes?
17.1 Exothermic and Endothermic Processes • In an endothermic process, the system gains heat as the surroundings cool down. • In an exothermic process, the system loses heat as the surroundings heat up.
17.1 Exothermic and Endothermic Processes • In studying energy changes, you can define a system as the part of the universe on which you focus your attention. The surroundings include everything else in the universe. • The law of conservation of energy states that in any chemical or physical process, energy is neither created nor destroyed.
17.1 Exothermic and Endothermic Processes • An endothermic process is one that absorbs heat from the surroundings.
17.1 Exothermic and Endothermic Processes • An exothermic process is one that releases heat to its surroundings.
A. Exothermic B. Endothermic ___1. making ice cubes ___2. melting ice cubes ___3. baking bread ___4. cooking an egg ___5. a candle flame ___6. condensation of rain from water vapor ___7. conversion of frost to water vapor
17.1 Heat Capacity andSpecific Heat • Water releases a lot of heat as it cools. During freezing weather, farmers protect citrus crops by spraying them with water.
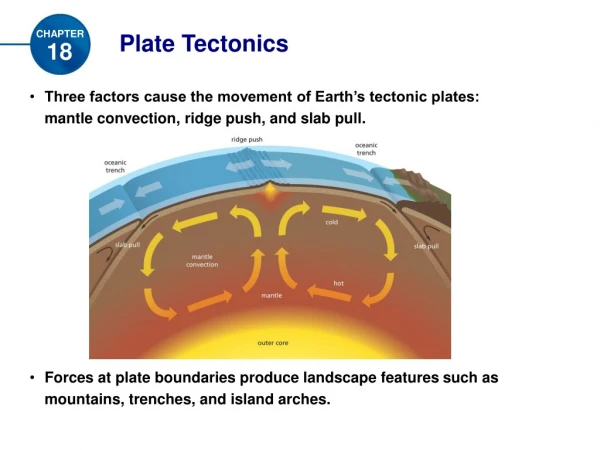
Thermodynamics BehindPlate Tectonics
The Plates and How They Move The lithosphere is broken into separate sections called plates. The theory of plate tectonics explains the formation, movement, and subduction of Earth’s plates.
Heat Transfer There are three types of heat transfer: radiation, conduction, and convection. Heat transfer – the movement of energy from a warmer object to a cooler object Conduction – heat transfer by direct contact of particles of matter Radiation – the transfer of energy through empty space
Convection & Convection Currents Heating and cooling of the fluid, changes in the fluid’s density, and the force of gravity combine to set convection currents in motion. Convection currents – the flow that transfers heat within a fluid Convection – heat transfer by the movement of a heated fluid
Convection Currents in Earth Heat from the core and the mantle causes convection currents in the mantle. This is how the heat is transferred, and how the earth’s plates are able to move.
Heat The higher the temperature, the faster the particles (atoms/molecules) are moving, i.e. more Kinetic Energy. We will take heat to mean the thermal energy in a body OR the thermal energy transferred into/out of a body
First law of Thermodymanics • The change in internal energy of a system equals the sum of the heat and work. • Energy can neither be created or destroyed.
Model Heat Engine (what goes in must come out)
Second Law of Thermodynamics • Energy transforms form one form to another. • The universe tend to favor entropy or disorder.