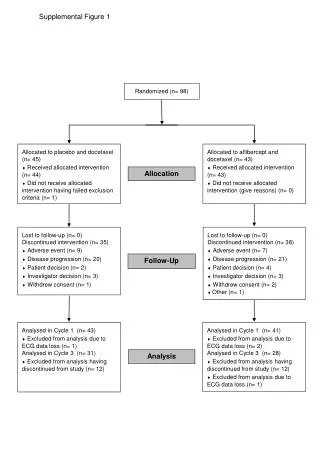
Clinical Study Overview: Intervention Allocation and Follow-Up Analysis in Cancer Treatment
This clinical study focuses on the allocation of patients to two groups: placebo with docetaxel and aflibercept with docetaxel. Among participants, 88% were randomized, with 44 receiving the allocated intervention for placebo, and all 43 for aflibercept. Discontinuation reasons included adverse events (9%), disease progression (20%), and patient/investigator decisions. Follow-up analysis was conducted in cycles 1 and 3, noting exclusions due to ECG data loss. These insights could guide future cancer treatment protocols and patient management strategies.


Clinical Study Overview: Intervention Allocation and Follow-Up Analysis in Cancer Treatment
E N D
Presentation Transcript
Randomized (n= 88) Allocated to placebo and docetaxel (n= 45) ¨Received allocated intervention (n= 44) ¨Did not receive allocated intervention having failed exclusion criteria (n= 1) Allocated to aflibercept and docetaxel (n= 43) ¨Received allocated intervention (n= 43) ¨Did not receive allocated intervention (give reasons) (n= 0) Allocation Lost to follow-up (n= 0) Discontinued intervention (n= 35) ¨Adverse event (n= 9) ¨Disease progression (n= 20) ¨Patient decision (n= 2) ¨Investigator decision (n= 3) ¨Withdrew consent (n= 1) • Lost to follow-up (n= 0) • Discontinued intervention (n= 38) • ¨Adverse event (n= 7) • ¨Disease progression (n= 21) • ¨Patient decision (n= 4) • ¨Investigator decision (n= 3) • ¨Withdrew consent (n= 2) • ¨ Other (n= 1) Follow-Up Analysed in Cycle 1 (n= 43)¨Excluded from analysis due to ECG data loss (n= 1) Analysed in Cycle 3 (n= 31) ¨Excluded from analysis having discontinued from study (n= 12) Analysed in Cycle 1 (n= 41)¨Excluded from analysis due to ECG data loss (n= 2) Analysed in Cycle 3 (n= 28) ¨Excluded from analysis having discontinued from study (n= 12) ¨Excluded from analysis due to ECG data loss (n= 1) Analysis Supplemental Figure 1