Chapter 3 Crystal Geometry and Structure Determination
“There are two things to aim at in life: first, to get what you want; and, after that, to enjoy it. Only the wisest of mankind achieve the second.” Logan Pearsall Smith, Afterthought (1931), “Life and Human Nature”. Chapter 3 Crystal Geometry and Structure Determination. Contents. Crystal.

Chapter 3 Crystal Geometry and Structure Determination
E N D
Presentation Transcript
“There are two things to aim at in life: first, to get what you want; and, after that, to enjoy it. Only the wisest of mankind achieve the second.”Logan Pearsall Smith, Afterthought (1931), “Life and Human Nature”
Contents Crystal Crystal, Lattice and Motif Symmetry Crystal systems Bravais lattices Miller Indices Structure Determination
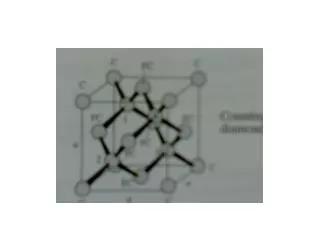
Crystal ? A 3D translationaly periodic arrangement of atoms in space is called a crystal.
Unit cell description : 1 Translational Periodicity One can select a small volume of the crystal which by periodic repetition generates the entire crystal (without overlaps or gaps) Unit Cell
UNIT CELL: Unit cell description : 2 The most common shape of a unit cell is a parallelopiped.
Unit cell description : 3 The description of a unit cell requires: 1. Its Size and shape (lattice parameters) 2. Its atomic content (fractional coordinates)
a c b Unit cell description : 4 Size and shape of the unit cell: 1. A corner as origin 2. Three edge vectors {a, b, c} from the origin define a CRSYTALLOGRAPHIC COORDINATE SYSTEM 3. The three lengths a, b, c and the three interaxial angles , , are called the LATTICE PARAMETERS
Lattice? A 3D translationally periodic arrangement of points in space is called a lattice.
Lattice A 3D translationally periodic arrangement of points Each lattice point in a lattice has identical neighbourhood of other lattice points.
Classification of lattice The Seven Crystal System And The Fourteen Bravais Lattices
7 Crystal Systems and 14 Bravais Lattices • Crystal System Bravais Lattices • Cubic P I F • Tetragonal P I • Orthorhombic P I F C • Hexagonal P • Trigonal P • Monoclinic P C • Triclinic P P: Simple; I: body-centred; F: Face-centred; C: End-centred
The three cubic Bravais lattices • Crystal system Bravais lattices • Cubic P I F Simple cubicPrimitive cubicCubic P Body-centred cubicCubic I Face-centred cubicCubic F
Orthorhombic CEnd-centred orthorhombicBase-centred orthorhombic
Cubic Crystals? a=b=c; ===90
7 crystal Systems Unit Cell Shape Crystal System 1. a=b=c, ===90 Cubic 2. a=bc,===90 Tetragonal 3. abc, ===90 Orthorhombic 4. a=bc, == 90, =120 Hexagonal 5. a=b=c, ==90 Rhombohedral OR Trigonal 6. abc, ==90 Monoclinic 7. abc, Triclinic
? Why half the boxes are empty? • Crystal System Bravais Lattices • Cubic P I F • Tetragonal P I • Orthorhombic P I F C • Hexagonal P • Trigonal P • Monoclinic P C • Triclinic P E.g. Why cubic C is absent?
End-centred cubic not in the Bravais list ? End-centred cubic = Simple Tetragonal
14 Bravais lattices divided into seven crystal systems • Crystal system Bravais lattices • Cubic P I F C • Tetragonal P I • Orthorhombic P I F C • Hexagonal P • Trigonal P • Monoclinic P C • Triclinic P
Now apply the same procedure to the FCC lattice Cubic F = Tetragonal I ?!!!
14 Bravais lattices divided into seven crystal systems • Crystal system Bravais lattices • Cubic P I F C • Tetragonal P I • Orthorhombic P I F C • Hexagonal P • Trigonal P • Monoclinic P C • Triclinic P
ML Frankenheim Auguste Bravais 1801-1869 1811-1863 1835: 15 lattices 1850: 14 lattices Couldn’t find his photo on the net History: IIT-Delhi 26th July 2013: AML120 Class Sem I 2013-2014 13 lattices !!! X 1856: 14 lattices
UNIT CELLS OF A LATTICE If the lattice points are only at the corners, the unit cell is primitive otherwise non-primitive Non-primitive cell Primitivecell A unit cell of a lattice is NOT unique. Primitivecell Unit cell shape CANNOT be the basis for classification of Lattices
A good after-class question from the last class: UNIT CELLS OF A LATTICE If we are selecting smallest possible region as a unit cell, why can’t we select a triangular unit cell? Unit cell is a small volume of the crystal which by periodic repetition generates the entire crystal (without overlaps or gaps) Primitivecell
Why can’t the Face-Centred Cubic lattice (Cubic F) be considered as a Body-Centred Tetragonal lattice (Tetragonal I) ?
What is the basis for classification of lattices into 7 crystal systems and 14 Bravais lattices?
Symmetry? If an object is brought into self-coincidence after some operation it said to possess symmetry with respect to that operation.
Translational symmetry Lattices also have translational symmetry In fact this is the defining symmetry of a lattice
Rotation Axis If an object come into self-coincidence through smallest non-zero rotation angle of then it is said to have an n-fold rotation axis where =180 2-fold rotation axis n=2 n=4 4-fold rotation axis =90
Rotational Symmetries Z Angles: 180 120 90 72 60 45 Fold: 6 2 3 4 5 8 Graphic symbols
Crsytallographic Restriction 5-fold symmetry or Pentagonal symmetry is not possible for Periodic Tilings Symmetries higher than 6-fold also not possible Only possible rotational symmetries for lattices 2 3 4 5 6 7 8 9…
Symmetry of lattices Lattices have Translational symmetry Rotational symmetry Reflection symmetry
Point Group and Space Group The group of all symmetry elements of a crystal except translations (e.g. rotation, reflection etc.) is called its POINT GROUP. The complete group of all symmetry elements including translations of a crystal is called its SPACE GROUP
Classification of Lattices Classification of lattices Crystal systems and Bravais Lattices Based on the point group symmetry alone (i.e. excluding translational symmetry 7 types of lattices 7 crystal systems Based on the space group symmetry, i.e., rotational, reflection and translational symmetry 14 types of lattices 14 Bravais lattices
7 crystal Systems Defining Crystal system Conventionalsymmetryunit cell a=b=c, ===90 4 Cubic a=bc,===90 A single Tetragonal abc, ===90 3 Orthorhombic a=bc, == 90, =120 Hexagonal 1 a=b=c, ==90 A single Rhombohedral abc, ==90 A single Monoclinic abc, none Triclinic
Tetragonal symmetry Cubic symmetry Cubic C = Tetragonal P Cubic F Tetragonal I
End of Lec4 on 30.07.2013 Lec2 on crystallography
Crystal Lattice A 3D translationally periodic arrangement of atoms A 3D translationally periodic arrangement of points
What is the relation between the two? Crystal = Lattice + Motif Motif or basis: an atom or a group of atoms associated with each lattice point
Crystal=lattice+basis Lattice: the underlying periodicity of the crystal, Basis: atom or group of atoms associated with each lattice points Lattice: how to repeat Motif: what to repeat
Lattice + Motif = Crystal = + Love Lattice + Heart = Love Pattern
Air, Water and Earth by M.C. Esher
Every periodic pattern (and hence a crystal) has a unique lattice associated with it
The six lattice parameters a, b, c, , , The cell of the lattice lattice + Motif crystal
Richard P. Feynman Nobel Prize in Physics, 1965
Feynman’s Lectures on Physics Vol 1 Chap 1 Fig. 1-4 Hexagonal symmetry “Fig. 1-4 is an invented arrangement for ice, and although it contains many of the correct features of the ice, it is not the true arrangement. One of the correct features is that there is a part of the symmetry that is hexagonal. You can see that if we turn the picture around an axis by 120°, the picture returns to itself.”
Correction: Shift the box One suggested correction: But gives H:O = 1.5 : 1 http://www.youtube.com/watch?v=kUuDG6VJYgA