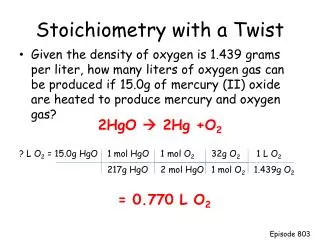Stoichiometry with two givens
140 likes | 363 Vues
Stoichiometry with two givens. By Erica Dougherty. If 17.65 g of aluminum oxide is reacted with 25.42 g of bromine, how many grams of aluminum bromide is produced? What is the excess reagent? What is the limiting reagent? How much of the excess reagent is left over?.

Stoichiometry with two givens
E N D
Presentation Transcript
Stoichiometry with two givens By Erica Dougherty
If 17.65 g of aluminum oxide is reacted with 25.42 g of bromine, how many grams of aluminum bromide is produced? What is the excess reagent? What is the limiting reagent? How much of the excess reagent is left over?
Write a complete and balanced equation. 2Al2O3 + 6Br2 --------> 4AlBr3 + 3O2
Draw columns after each chemical, and divide your paper in half. 2Al2O3 + 6Br2 --------> 4AlBr3 + 3O2
Write the amounts given in the proper columns, make sure one is on the top level and the other is on the bottom level. 2Al2O3 + 6Br2 --------> 4AlBr3 + 3O2 17.65g
Convert the given into moles. 2Al2O3 + 6Br2 --------> 4AlBr3 + 3O2 17.65g*1mole/101.961= .1731moles
In each of the other columns, write moles given in each of the levels times a fraction. 2Al2O3 + 6Br2 --------> 4AlBr3 + 3O2 17.65g*1mole/101.961= .1731moles .1731* / .1731* / .1731* /
The numerator of the fraction is the coefficient of that column. 2Al2O3 + 6Br2 --------> 4AlBr3 + 3O2 17.65g*1mole/101.961= .1731moles .1731* 6/ .1731* 4/ .1731* 3/
The denominator of the fraction is the coefficient of the given column on the same level. (top or bottom) 2Al2O3 + 6Br2 --------> 4AlBr3 + 3O2 17.65g*1mole/101.961= .1731moles .1731* 6/2 .1731* 4/2 .1731* 3/2
Do math to find moles. 2Al2O3 + 6Br2 --------> 4AlBr3 + 3O2 17.65g*1mole/101.961= .1731moles .1731* 6/2= .5193moles .1731* 4/2= .3462moles .1731* 3/2= .2597moles
Compare moles of one element. Cross off the row with more moles. 2Al2O3 + 6Br2 --------> 4AlBr3 + 3O2 17.65g*1mole/101.961= .1731moles .1731* 6/2= .5193moles .1731* 4/2= .3462moles .1731* 3/2= .2597moles
Convert moles into grams. 2Al2O3 + 6Br2 --------> 4AlBr3 + 3O2 17.65g*1mole/101.961= .1731moles .1731* 6/2= .5193moles .1731* 4/2= .3462moles .1731* 3/2= .2597moles
Verify law of conservation of mass. 2Al2O3 + 6Br2 --------> 4AlBr3 + 3O2 17.65g*1mole/101.961= .1731moles .1731* 6/2= .5193moles .1731* 4/2= .3462moles .1731* 3/2= .2597moles
Answers • 28.30g of aluminum bromide are produced. • Aluminum oxide is the excess reagent. • Bromine is the limiting reagent. • There is 12.25g of aluminum oxide left.