CHAPTER 7-A Light and Atomic Spectra
CHAPTER 7-A Light and Atomic Spectra. The Periodic Table. The periodic table is the most important organizing principle in chemistry. Chemical and physical properties of elements in the same group are similar.


CHAPTER 7-A Light and Atomic Spectra
E N D
Presentation Transcript
CHAPTER 7-A Light and Atomic Spectra © 2012 by W. W. Norton & Company
The Periodic Table • The periodic table is the most important organizing principle in chemistry. • Chemical and physical properties of elements in the same group are similar. • All chemical and physical properties vary in a periodic manner, hence the name periodic table.
Electromagnetic Radiation Electromagnetic Radiation: Energy propagated by an electromagnetic field. Electromagnetic radiation has both particle and wave nature.
Electromagnetic Radiation Spectroscopy: Branch of physical science that deals with the interaction of electromagnetic radiation with matter Spectrometry: The quantitative measurement of the intensity of radiation at a particular wavelength of light.
Wave-Like Nature of Light Frequency (, Greek nu):Number of peaks that pass a given point per unit time. Wavelength (, Greek lambda):Distance from one wave peak to the next. Amplitude:Height measured from the center of the wave. The square of the amplitude givesintensity.
Wave-Like Nature of Light • Speedof a wave is the wavelength (in meters) multiplied by its frequency in reciprocal seconds. • Wavelength x Frequency = Speed • (m) x (s–1) = c (m/s)
Particle-Like Nature of Light Electromagnetic radiation can be described as a stream of tiny particles, called photons, with a very small mass and a very large velocity. The velocity of photons traveling in a vacuum is: c = 3.00 x 108 m/s
Particle-Like Nature of Light Where does a photon come from? One photon is emitted when one atom or molecule in an excited state relaxes to the ground state via the emission of radiation. E = h ν
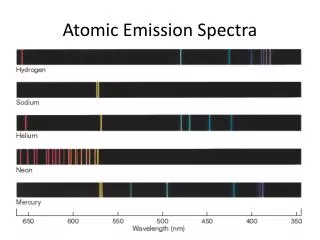
Atomic Spectra • Atomic spectra: Result from excited atoms emitting light. • Line spectra:Result from electron transitions between specific energy levels.