Understanding Organic Halides: Functional Groups, Nomenclature, and Reactivity
This text provides an overview of organic halides, describing their functional groups as structural units responsible for characteristic behaviors under specific reaction conditions. It covers the nomenclature of alkyl halides using IUPAC methods, emphasizing both functional class and substitutive nomenclature. The document discusses the types of alkyl halides, including primary and secondary forms, and delves into aryl halides and their properties. Additionally, it examines the physical properties and boiling points of various halide compounds, as well as the reactivity of alcohols when treated with hydrogen halides.

Understanding Organic Halides: Functional Groups, Nomenclature, and Reactivity
E N D
Presentation Transcript
Functional Group • A structural unit in a molecule responsible for itscharacteristic behavior under a particular set ofreaction conditions
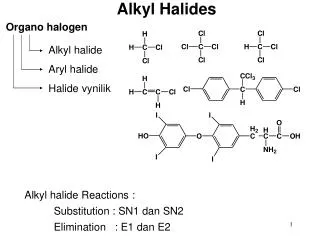
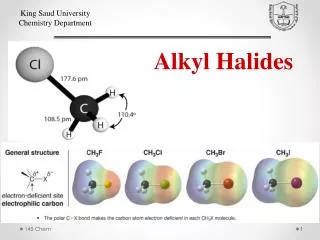
Families of organic compoundsand their functional groups • Alcohol ROH • Alkyl halide RX (X = F, Cl, Br, I) • Amine primary amine: RNH2 • secondary amine: R2NH • tertiary amine: R3N
IUPAC Nomenclature There are several kinds of IUPAC nomenclature. • The two that are most widely used are:functional class nomenclaturesubstitutive nomenclature • Both types can be applied to alcohols andalkyl halides.
CH3CH2CHCH2CH2CH3 H I Br Functional Class Nomenclature of Alkyl Halides • Name the alkyl group and the halogen asseparate words (alkyl + halide) CH3CH2CH2CH2CH2Cl CH3F
CH3CH2CHCH2CH2CH3 H I Br Functional Class Nomenclature of Alkyl Halides • Name the alkyl group and the halogen asseparate words (alkyl + halide) CH3F CH3CH2CH2CH2CH2Cl Methyl fluoride Pentyl chloride 1-Ethylbutyl bromide Cyclohexyl iodide
CH3CHCH2CH2CH3 Br CH3CH2CHCH2CH3 I Substitutive Nomenclature of Alkyl Halides • Name as halo-substituted alkanes. • Number the longest chain containing thehalogen in the direction that gives the lowestnumber to the substituted carbon. CH3CH2CH2CH2CH2F
CH3CHCH2CH2CH3 Br CH3CH2CHCH2CH3 I Substitutive Nomenclature of Alkyl Halides • Name as halo-substituted alkanes. • Number the longest chain containing thehalogen in the direction that gives the lowestnumber to the substituted carbon. CH3CH2CH2CH2CH2F 1-Fluoropentane 2-Bromopentane 3-Iodopentane
Cl CH3 CH3 Cl Substitutive Nomenclature of Alkyl Halides • Halogen and alkyl groupsare of equal rank when it comes to numberingthe chain. • Number the chain in thedirection that gives the lowest number to thegroup (halogen or alkyl)that appears first.
Cl CH3 CH3 Cl Substitutive Nomenclature of Alkyl Halides 5-Chloro-2-methylheptane 2-Chloro-5-methylheptane
Nomenclature of Alkyl Halides In the IUPAC system, alkyl halides are named as substituted alkanes
CH3 CH3CHCH2CH2CH3 CH3CCH2CH2CH3 Br OH Classification H CH3CH2CH2CH2CH2F OH primary alkyl halide secondary alcohol secondary alkyl halide tertiary alcohol
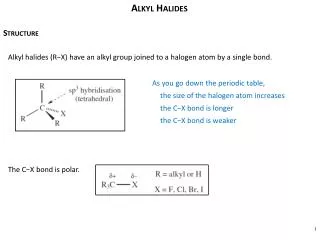
Aryl Halides • Aryl halides are halides in which the halogen is attached directly to an aromatic ring. • Carbon-halogen bonds in aryl halides are shorter and stronger than carbon-halogen bonds in alkyl halides.
Aryl Halides • Aryl halides are halides in which the halogen is attached directly to an aromatic ring. • Carbon-halogen bonds in aryl halides are shorter and stronger than carbon-halogen bonds in alkyl halides. • Because the carbon-halogen bond is stronger, aryl halides react more slowly than alkyl halides when carbon-halogen bond breaking is rate determining.
Boiling point increases with increasingnumber of halogens Compound Boiling Point • CH3Cl -24°C • CH2Cl2 40°C • CHCl3 61°C • CCl4 77°C Even though CCl4 is the only compound in this list without a dipole moment, it has the highest boiling point. Induced dipole-induced dipole forces are greatest in CCl4 because it has the greatest number of Cl atoms. Cl is more polarizable than H.
Cl Cl Physical Properties of Aryl Halides • resemble alkyl halides • all are essentially insoluble in water • less polar than alkyl halides 2.2 D 1.7 D
least reactive most reactive Reaction of Alcohols with Hydrogen Halides ROH + HX RX + HOH • Hydrogen halide reactivity HF HCl HBr HI
An SN2 reaction proceeds in the direction that allows the strongest base to displace the weaker base
1. NaOH, H2O 370°C Cl OH 2. H+ Example (97%)
Cl OCH3 NO2 NO2 But... • nitro-substituted aryl halides do undergonucleophilic aromatic substitution readily CH3OH + + NaOCH3 NaCl 85°C (92%)
F OCH3 NO2 NO2 Reaction CH3OH + + NaOCH3 NaF 85°C (93%)
Cl NH2 Aryl Halides Undergo Substitution WhenTreated With Very Strong Bases KNH2, NH3 –33°C (52%)
* Cl OH * OH * Hydrolysis of Chlorobenzene • 14C labeling indicates that the high-temperature reaction of chlorobenzene with NaOH goes via benzyne. NaOH, H2O 395°C + (43%) (54%)