Energy Transformations
Energy Transformations. Georgia High School Graduation Test: Science Review Mrs. Kirby. Introduction. From the key vocabulary, circle the words that you can already define or use in a sentence. Write down two or three things that you think are important for you to know today.

Energy Transformations
E N D
Presentation Transcript
Energy Transformations Georgia High School Graduation Test: Science Review Mrs. Kirby
Introduction • From the key vocabulary, circle the words that you can already define or use in a sentence. • Write down two or three things that you think are important for you to know today.
Sample Question • Describe the process of radioactive decay in which the unstable nucleus of a radioactive isotope spontaneously decays.
Radioactivity • the spontaneous process that occurs when an unstable nucleus releases particles and/or energy to form a more stable nucleus
Radioisotopes • unstable radioactive nucleus • examples: • carbon-14 • deuterium (hydrogen with 1 neutron) • all elements with atomic numbers above 83
Radioactive Decay • radioactive decay = the release of subatomic particles by radioactive isotopes • alpha and beta decay result in transformed atom • gamma decay results in same atom with release of energy ALPHA DECAY BETA DECAY GAMMA DECAY
Sample Question • Calculate the amount of radioactive substance that will remain after one half-life.
Half-life • the time it takes for half of the radioactive particles in a radioactive nucleus to decay • example: • If an original sample of carbon-14 has a mass of 10 grams and its half-life is 5700 years, how many grams of carbon-14 would remain after one half-life? • ANSWER: 5 grams
Sample Question • What is the effect of an increase in temperature on the motion of atoms?
Temperature • a measure of the average kinetic energy of molecules • the higher the temperature, the faster the molecules
Sample Question • Compare solids, liquids, and gases in terms of shape, volume, and movement of particles.
Sample Question • What is the effect of temperature change on the phases of matter?
Phase Changes • to increase the temperature of a substance energy must be absorbed by that substance • to decrease the temperature of a substance energy must be released by that substance
Sample Question • Give examples and describe the two forms of energy encountered in a system between any of the following: chemical, heat, light, electrical, and mechanical.
Types of Energy • potential energy = stored energy due to position or chemical composition • kinetic energy = energy due to motion
Heat Energy • Heat can be transferred through: • Conduction = when objects touch • Convection = when matter moves • Radiation = in the form of waves (does not require matter) • Conductors = easily transmit energy • Example: metals • Insulators = do not easily transmit energy • Example: gases such as air
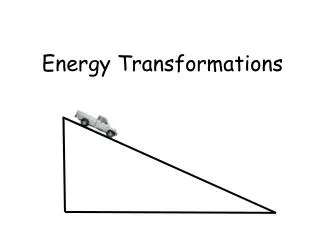
Conversion of Energy • The sum of KE and PE is constant if there is no friction. • Realistically, some energy is transferred to heat due to the friction between the wheels and the track.
Discussion • The molecular theory of molecules states that the velocity of molecules correlates to the temperature of the system. How are the actions of a group of people affected by the temperature of a room? How are those actions similar to and different from atoms at the same temperature?
Lesson Summarized • Write a sentence that explains the system discussed. • Draw a graphic organizer that shows the relationship of the parts to the whole.
Short Quiz Answers • Half-time is the time it takes for half of the radioactive particles in a radioactive nucleus to decay. • All isotopes have the same atomic number but different atomic masses. • Some isotopes are radioactive due to the different number of neutrons. • See the table on sources of energy. • Ice, water, and steam all have the same chemical composition (H2O) but each are in a different phase.
More Practice • From the blue book, complete the following questions: • p.53 (26,27,30) • p.61 (1-4) • p.62-63 (6-8) • p.64 (11-14) • p.68 (21-25)