The Atom
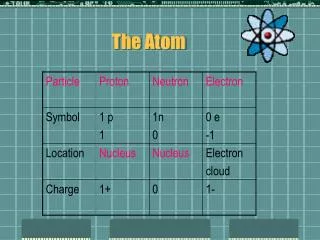
The Atom. What is an Atom?. Atom : Smallest unit of all matter 3 Main Parts: 1) Proton (+ charge) 2) Neutron (No charge) 3) Electron (- charge) Protons & Electrons usually equal in number Elements (substance made of only 1 type of atom) are identified on the Periodic Table.

The Atom
E N D
Presentation Transcript
What is an Atom? • Atom: Smallest unit of all matter • 3 Main Parts: • 1) Proton (+ charge) • 2) Neutron (No charge) • 3) Electron (- charge) • Protons & Electrons usually equal in number • Elements (substance made of only 1 type of atom) are identified on the Periodic Table
Atomic Number: Number of protons the atom contains Usually # of electrons also Atomic Mass: Combined # of protons and neutrons inside the nucleus Electrons too small to affect mass How many protons, electrons, and neutrons does Carbon contain? Protons = ______ Electrons = ______ Neutrons = ______ Reading the Periodic Table 6 6 6
What’s the: 4 • Atomic number of Beryllium? B) Atomic mass of Beryllium? C) How many protons? D) How many electrons? E) How many neutrons? 9 4 4 5
Electron Cloud Examine Carbon below. It has 2 electrons in the first level and 4 electrons in the 2nd level. Is Carbon stable? • Electrons orbit the nucleus in “energy levels” 1st level: • closest to nucleus • stable w/ 2 electrons 2nd level: • stable w/ 8 electrons 3rd level: • stable w/ 8 electrons • Atoms are stable when the outermost level is “full”
Molecules • Molecule: 2 or more atoms held together by a covalent bond • Form when atoms are unstable • Covalent Bond: Chemical bond where atoms share electrons • Ex: Oxygen atom • Why unstable? Only 6 electrons in outer energy level • What happens? Each oxygen atom will share two electrons with the other oxygen • End result? Both will have 8 electrons in its outer level (O2 is created)
Ionic Bonds • Ionic bond: chemical bond where electrons are gained/lost • Creates Ions (atoms with + or – charge) • Held together by magnetic attraction • Cause: 1 atom steals an electron(s) from another atom • Ex: Sodium + Chlorine = Sodium Chloride sodium chlorine
Ionic Bonds e e e e e e e e e e e e e sodium e e e e e e chlorine e e e e e e e e e After bonding Before bonding 11+ 11+ 17+ 17+ 10- 18- 11- 17- +1 -1 0 0
Kobe Kuiz • Name the 3 subatomic particles. • Which subatomic particles are found inside the nucleus? • The atomic number usually allows us to determine the amount of which two subatomic particles? • If an atom has the atomic mass of 14 and the atomic number of 6, how many protons, neutrons, and electrons does the atom contain? • After an ionic bond is formed, what is the charge of the atom that gained an electron? • Which type of chemical bond shares electrons?