NUCLEIC ACIDS
NUCLEIC ACIDS. DNA RNA. DNA(Deoxyribo nucleic acid ) RNA (Ribo nucleic acid ). Brief History of Nucleotides and Nucleic acids. 1869 – Johannes Friedrich Miescher isolated “nuclein” from soiled bandages.

NUCLEIC ACIDS
E N D
Presentation Transcript
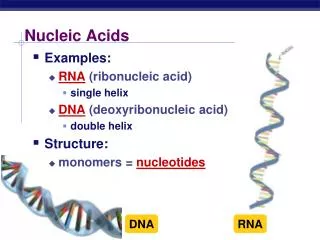
NUCLEIC ACIDS DNA RNA DNA(Deoxyribonucleic acid) RNA (Ribonucleic acid)
Brief History of Nucleotidesand Nucleic acids • 1869 – Johannes FriedrichMiescher isolated “nuclein” from soiled bandages. • 1902 – ArchibaldGarrod studied “Alkaptonuria”; he also concluded that specific gene is associated with absence of specific enzyme. • 1903 – WalterSutton, introduced the chromosome structure. • 1913 – Thomas HuntMorgan “gene mapping” • 1926 – JamesSumner purified urease and he also identified enzymes to be proteins.
Brief History of Nucleotidesand Nucleic acids • 1928 –Frederick Griffith “Transforming Principle” a chemical transferred from dead bacteria to living cells caused genetically converted strains. • 1944 – Oswald Avery, Maclyn McCarty, and Colin MacLeod – identified Griffith’s “transformation principle” as DNA. • 1947 - Erwin Chargaff – studied the base pairing • 1950’s – Rosalind Franklin “X-ray of DNA”
Brief History of Nucleotidesand Nucleic acids 1953: James Watsons and Francis Crick – “DNA double helix”. "Molecular structure of Nucleic Acids: A Structure for Deoxyribose Nucleic Acid" An article published in Nature (April 25, 1953). It was the first publication which described the discovery of the double helix structure of DNA.
“Rungs of ladder” Nitrogenous Base (A,T,G or C) “Legs of ladder” Phosphate & Sugar Backbone Watson & Crick proposed... DNA double helix • DNA had specific pairing between the nitrogen bases: • ADENINE – THYMINE • CYTOSINE - GUANINE • DNA was made of 2 long stands of nucleotides arranged in a specific way called the “Complementary Rule”.
H-bonds G C A T BASE-PAIRINGS
THE WATSON-CRICK MODEL BASE-PAIRING IN DNA The two strands of DNA are arranged antiparallel to one another: viewed from left to right the "top" strand is aligned 5' to 3', while the "bottom" strand is aligned 3' to 5'.
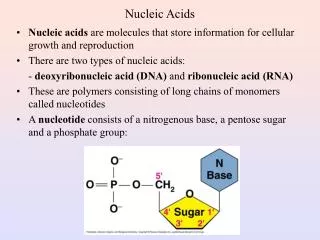
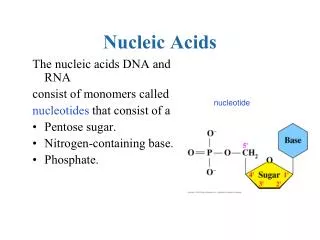
Nucleotides and Nucleic acids • are biological molecules that possess heterocyclic nitrogenous bases as principal components of their structure. • The biochemical roles of nucleotides: participate as essential intermediates in virtually all aspects of cellular metabolism. • Serving an even more central biological purpose are the nucleic acids, the elements of heredity and the agents of genetic information transfer. • Just as proteins are linear polymers of amino acids, _________ are linear polymers of nucleotides.
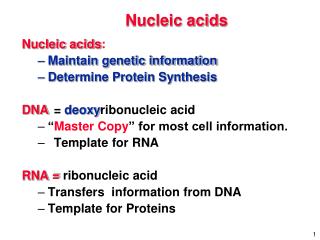
Nucleic acids are “Polymers” • Responsible for the storage and passage of the information needed for the production of proteins; • The chemical link between generations; • The source of genetic information in chromosomes; • Dictate amino-acid sequence in proteins; and • Give information to chromosomes, which is then passed from parent to offspring.
Nitrogenous Bases • The bases of nucleotides and nucleic acids are derivatives of either pyrimidine or purine. • Q: Look at the structure of pyrimidine, what can you observe? • A: 6 member heterocyclic ring, with 2 Nitrogen atoms and it is number clockwise. • Q: Look at the structure of purine, what can you observe? • A: It is 9 member ring structure of pyrimidine fused with an imidazole ring.
Common Pyrimidines and Purines 1. 2. 3. Q: What are the common pyrimidine bases? A: Cytosine, Uracil and Thymine Q: What is the name of the first pyrimidine base? What is its chemical name? Q: What is the name of the second pyrimidine base? What is its chemical name? Q: What is the name of the third pyrimidine base? What is its chemical name?
Common Pyrimidines and Purines 1. 2. Q: What are the common purine bases? A: Adenine and Guanine Q: What is the name of the first purine base? What is its chemical name? Q: What is the name of the first purine base? What is its chemical name?
Properties of Pyrimidines and Purines • keto-enoltautomeric shifts • pyrimidines and purines exist as tautomeric pairs • strong absorbance of ultraviolet (UV) light, which is also a consequence of the aromaticity of their heterocyclic ring structures.
Properties of Pyrimidines and Purines Hydrogen bonding between purine and pyrimidine bases is fundamental to the biological functions of nucleic acids, as in the formation of the double helix structure of DNA.
Properties of Pyrimidines and Purines • The important functional groups participating in H-bond formation: • amino groups of adenine, cytosine and guanine; • the ring nitrogens at position 3 of pyrimidines and position 1 of purines; and • the strongly electronegative oxygen atoms attached at position 4 of uracil and thymine, position 2 of cytosine, and position 6 of guanine
Pentose Sugar Five-carbon sugars are called _____. Q: What is the pentose sugar present in DNA? Q: What is the pentose sugar present in RNA?
Pentose Sugar • Pentose is in the five-membered ring form known as _______. • Q: What is the furanose present in DNA? • A: 2-deoxy-D-ribofuranose • Q: What is the furanose present in RNA? • A: D-ribofuranose
Nucleosides + Nucleosides are compounds formed when a base is linked to a sugar via a glycosidic bond. A glycosidic bond is a type of covalent bond that joins a sugar molecule to another group. Q: What is the name of the nucleoside if the sugar found is ribose? Q: What is the name of the nucleoside if the sugar found is 2-deoxyribose?
Nucleosides In nucleosides, the bond is an N-glycoside because it connects the anomeric C-1' to N-1 of a pyrimidine or to N-9 of a purine.
Nucleoside Nomenclature Nucleosides are named by adding the ending -idine to the root name of a pyrimidine or -osine to the root name of a purine.
Nucleoside Conformation syn and anti rotation of the base about the glycosidic bond is sterically hindered, principally by the hydrogen atom on the C-2' carbon of the furanose Pyrimidine nucleosides favor the anti conformation Purine nucleosides can adopt either the syn or anti conformation.
Nucleosides Are More Water-Soluble Than Free Bases Nucleosides are much more water-soluble than the free bases because of the hydrophilicity of the sugar moiety. Nucleosides are relatively stable in alkali. Pyrimidine nucleosides are also resistant to acid hydrolysis, but purine nucleosides are easily hydrolyzed in acid to yield the free base and pentose.
Nucleotides Are Nucleoside Phosphates • A nucleotide results when phosphoric acid is esterified to a sugar —OH group of a nucleoside. The nucleoside ribose ring has three —OH groups available for esterification, at C-2', C-3', and C-5'(although 2'-deoxyribose has only two). The vast majority of monomeric nucleotides in the cell are ribonucleotides having 5'-phosphate groups.
Nomenclature of Nucleotide Q: What are the names of this structure? Q: What are the names of this structure? Answers: 1.) adenosine 5’-monophosphate 2.) AMP or 5’-AMP 3.) adenylic acid 4.) adenylate Answers: 1.) guanosine 5’-monophosphate 2.) GMP or 5’-GMP 3.) guanylic acid 4.) guanylate
Nomenclature of Nucleotide Q: What are the names of this structure? Q: What are the names of this structure? Answers: 1.) cytidine 5’-monophosphate 2.) CMP 3.) cytidylic acid 4.) cytidylate Answers: 1.) uridine 5’-monophosphate 2.) UMP 3.) uridylic acid 4.) uridylate
Cyclic Nucleotides Nucleoside monophosphates in which the phosphoric acid is esterified to two of the available ribose hydroxyl groups are found in all cells. Forming two such ester linkages with one phosphate results in a cyclic structure.3',5'-cyclic AMP, often abbreviated cAMP, and its guanine analog 3',5'-cyclic GMP, or cGMP, are important regulators of cellular metabolism.
Nucleoside 5'-Triphosphates Are Carriers of Chemical Energy ATP has been termed the energy currency of the cell. GTP is the major energy source for protein synthesis. CTP is an essential metabolite in phospholipid synthesis UTP forms activated intermediates with sugars that go on to serve as substrates in the biosynthesis of complex carbohydrates and polysaccharides.
Nucleoside 5'-Triphosphates Are Carriers of Chemical Energy The evolution of metabolism has led to the dedication of one of these four NTPs to each of the major branches of metabolism. To complete the picture, the four NTPs and their dNTP counterparts are the substrates for the synthesis of the remaining great class of biomolecules—the nucleic acids.
The Bases of Nucleotides Serve as “Information Symbols” ATP: serve as the primary nucleotide in central pathways of energy metabolism, GTP: is used to drive protein synthesis. Various nucleotides are channeled in appropriate metabolic directions through specific recognition of the base of the nucleotide. This role as information symbols extends to nucleotide polymers, the nucleic acids, where the bases serve as the information symbols for the code of genetic information.
Activity Identify the components of this structure. Classify the structure as that of a nucleoside or a nucleotide. Identify the nitrogenous whether a purine or pyrimidine. What is the sugar component in the structure? What is the name of the type of bond that occurs between a phosphate and ribose group? Between a ribose and nitrogen base group? Name the bond that forms between nucleotide groups. Explain the significance of hydrogen bonds in DNA helices.
Solution to the Activity • The structure contains a nitrogenous base and a pentose. • It is a nucleoside, because it contains a pentose and a nitrogenous base. • The nitrogenous base is pyrimidine. Cytosine. • The sugar has a -H in the 2' position and is therefore deoxyribose.
Solution to the Activity An ester bond forms between a phosphate group and a ribose group. A glycosidic bond forms between a ribose group and a nitrogen base group A phosphodiester bond forms between nucleotides to form nucleic acids. While hydrogen bonds do contribute a small amount to the stability of helices, their main contribution is to the specificity of a helix. Hydrogen bonds dictate the complementary base pairing that aligns anti-parallel nucleic acids strands in a DNA helix.
Chemical Nature of DNA 1. 2. 3.
DNA at a glance... • DNA is organized into chromosomes, which are found within the nuclei of cells. • A gene is a segment of DNA on a chromosome that codes for specific protein and thus determines a trait. • The genetic code is determined by the order of bases in the gene, which specifies what type of proteins will be produced. • Remember this! DNA contains the genetic material.
DNA – Deoxyribonucleic acid Chemical basis of heredity and is organized into genes, the fundamental units of genetic information. FUNCTIONS: Involve in replication during cell division Gene expression by transcription
DNA – Deoxyribonucleic acid In eukaryotic cells, it is present in chromosomes in the nucleus It is also present in mitochondria and in the chloroplast of plants Prokaryotic cells, which lack nuclei, have a single chromosome but may also contain nonchromosomal DNA in the form of plasmid.
Structure of DNA • Polydeoxyribonucleotide • Linked by 3’,5’-phosphodiester bonds • Double stranded molecules • In eukaryotic cells: nucleoproteins • In prokaryotic cells: nucleoid
Structure of DNA • 3’,5’-phosphodiester bonds
Structure of DNA where the phosphate group of the previous nucleotide is linked • 3’,5’-phosphodiester bonds where the phosphate group of the next nucleotide in a series can be linked
Structure of DNA:Double Helix Common axis The chains are paired in anti-parallel manner
“Rungs of ladder” Nitrogenous Base (A,T,G or C) “Legs of ladder” Phosphate & Sugar Backbone Structure of DNA:Double Helix
Structure of DNA:Base Pairing Note: the hydrogen bonds plus the hydrophobic interactions between the stacked bases stabilize the structure of the double helix.
Separation of the two DNA strands in the double helix • Q: How the separation occurs? • A: The 2 strands of the double helix occur when the hydrogen bonds between the paired bases are disrupted. • Q: What is the mechanism of disruption? • A: The pH of the DNA solution is altered so that the nucleotide bases ionize or if the solution is heated. • Note: no phosphodiester bond bonds are broken by such treatment.