Chapter 3 Atomic Structure
Chapter 3 Atomic Structure. You will explain why the model of the atom changed throughout history. Objective:. What is a model?. Models are used to help us understand things that cannot be seen directly


Chapter 3 Atomic Structure
E N D
Presentation Transcript
You will explain why the model of the atom changed throughout history. Objective:
What is a model? • Models are used to help us understand things that cannot be seen directly • Models are used when something is too large, too small, or too dangerous to be studied directly.
Examples of Scientific Models This model shows the alignment between the sun, moon, & earth. As it rotates, it shows the phases of the moon and how we measure a year. This model is a mathematical representation of a sound wave. You cannot see sound, but you can see how it affects other objects with its vibrations.
Examples of Scientific Models • What other examples can you think of? • Are there other models present in this room?
A good model… • Must be based on observations and indirect experimentation. • Must explain as many characteristics of the original object as possible. • Should be as simple as possible.
When do you change a model? • All models have limitations —No model has ever been totally complete. • A model changes when observations of a new situation do not agree with the current model.
Creating a Model • The “Think Tube”is also a model for something you cannot see directly. ? THINK TUBE
Creating a Model • How many strings are on the inside? • Make your own model showing how the “Think Tube” works.
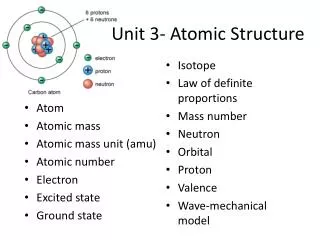
Atoms • The atom has not changed over time, but our idea and model of the atom has.
Definition of Atom: • the smallest particle of an element that retains the chemical properties of that element.
Democritus 400 BC By convention there is color,By convention sweetness,By convention bitterness,But in reality there are atoms and space. -Democritus (c. 400 BCE) Convention means because we said so- doesn’t really exist
Democritus 400 BC • Democritus was smashing up sea shells one day and thought that you can break down the shell to tiny pieces, but it can not be completely destroyed.
Democritus 400 BC • Looked at sand on the beach. Cut sand in half and got fewer and fewer grains of sand. • What was the smallest piece? • He called it atomos = greek word meaning cannot be cut
Democritus 400 BCAccording to Democritus atoms are: • Invisible • Indivisible • Solid • Eternal • Surrounded by an empty space http://www.brl.ntt.co.jp/group/butsuden-g/img/redball.gif
Democritus 400 BCcontinued: • Have an infinite number of possible • shapes. • Each type of atom had a different size.
Democritus 400 BCAtoms - http://www.brl.ntt.co.jp/group/butsuden-g/img/redball.gif
Aristotle’s Idea 300 BC • All substances are made of 4 elements: Fire, Air, Earth, and Water
Aristotle’s Idea 300 BC • There were also four qualities: dryness, hotness, coldness, and moistness. • Fire was dry and hot, while water was moist and cold, etc. • Each of these elements move naturally in a line to their "proper place," where it will be at rest.
Aristotle 300 BC • Water sits on top of the earth, he explained, because it is lighter, yet air floats above the water because it is lighter still—and fire, lightest of all, rises highest. Furthermore, he claimed that the planets beyond Earth were made up of a "fifth element," or quintessence, of which little could be known.
Democritus vs Aristotle • Ancient Greeks accepted Aristotle’s ideas and rejected Democritus. • What holds the particles together? • Democritus could not answer this question • Remained that way until the 17th century
Important Discoveries • Law of Conservation of Mass • Law of Definite Proportions • Law of Multiple Proportions
Law of Conservation of Mass (Matter) • Lavosier measured the mass of chemicals before and after a chemical reaction and found that the weight did not change. • In a chemical reaction, matter is neither created nor destroyed.
Law of Definite Proportions • Also called Law of Constant Composition • Proposed by Joseph Proust • Elements always react and combine with one another in the same proportions.
Law of Definite Proportions A chemical compound is always composed of the same combination of atoms - copper carbonate CuCO3 http://cwx.prenhall.com/bookbind/pubbooks/hillchem3/medialib/media_portfolio/text_images/CH02/FG02_01.JPG
Law of Definite Proportions • Water has the formula H2O. This means that water in the ocean, lakes, or in our sinks always contains 2 atoms of Hydrogen for every 1 atom of Oxygen. What percent of water is Hydrogen and what percent is Oxygen? H: 2g/18g = 11% O: 16g/18g = 89%
Law of Multiple Proportions • If two elements form more than one compound between them, then the ratios of the weights of the two atoms will be ratios that can be reduced to small whole numbers. http://cwx.prenhall.com/bookbind/pubbooks/hillchem3/medialib/media_portfolio/text_images/CH02/FG02_02.JPG
John Dalton 1766-1844 • English school teacher and public lecturer by the age of 12. • As a Quaker, Dalton led a modest existence, although he received many honors later in life. In tribute, more than 40,000 people marched in his funeral procession. http://www.unit5.org/christjs/John_Dalton.htm
John Dalton’s Atomic Theory • Dalton’s Theory was a return to the ideas of Democritus • Dalton turned the idea into a scientific theory that could be tested • Not all of Daltons ideas are still true today. Some ideas were modified.
John Dalton’s Atomic Theory: • All matter is composed of atoms. • Atoms of a particular element have identical properties. Elements of a different element have different properties. • Atoms cannot be divided or destroyed. • Atoms combine to form compounds. • During a chemical reaction atoms are rearranged.
John Dalton’s Atomic Theory: • All matter is composed of atoms. • Atoms of a particular element have identical properties. Elements of a different element have different properties. • Atoms cannot be divided or destroyed. • Atoms combine to form compounds. • During a chemical reaction atoms are rearranged.
JJ Thomson 1856-1940 Excuse me... how can you discover a particle so small that nobody has ever seen one? http://www.aip.org/history/electron/jjsound.htm
J.J. Thompson’s Model _ _ + + _ + + _ _ +
Discovery of the Electron JJ Thomson determined that: • Atom is a sphere of positive matter that holds electrons in it. • Also called the plum pudding model or the raisin dough model.
His experiment http://www.sciencemuseum.org.uk/on-line/electron/section2/shockwave2.asp
Voltage source Thomson’s Experiment - + Vacuum tube Metal Disks
Voltage source Thomson’s Experiment - +
Voltage source Thomson’s Experiment - +
Voltage source Thomson’s Experiment - +
Voltage source Thomson’s Experiment - +
Voltage source Thomson’s Experiment - +
Voltage source Thomson’s Experiment - +
Voltage source J.J. Thompson’s Cathode Ray Tube • The cathode ray travels from the cathode to the anode when current was passed through the tube. - + Cathode Anode
Voltage source Thomson’s Experiment + - • By adding an electric field
Voltage source Thomson’s Experiment + - • By adding an electric field
Voltage source Thomson’s Experiment + - • By adding an electric field