Potentiometric Methods
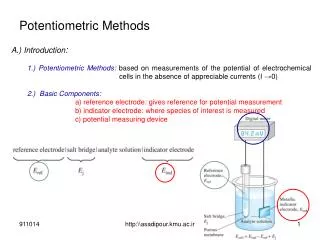
Potentiometric Methods. A.) Introduction : 1.) Potentiometric Methods: based on measurements of the potential of electrochemical cells in the absence of appreciable currents (I →0 ) 2.) Basic Components: a) reference electrode: gives reference for potential measurement

Potentiometric Methods
E N D
Presentation Transcript
Potentiometric Methods A.) Introduction: 1.) Potentiometric Methods: based on measurements of the potential of electrochemical cells in the absence of appreciable currents (I →0) 2.) Basic Components: a) reference electrode: gives reference for potential measurement b) indicator electrode: where species of interest is measured c) potential measuring device http:\\asadipour.kmu.ac.ir
Electrodes and Potentiometry • Potential change only dependent on one ½ cell concentrations • Reference electrode is fixed or saturated doesn’t change! Ecell=Ecathod-Eanod Anod is conventionally reference electode Fe3+ +e- Fe2+ AgCl(s) + e- → Ag + Cl- Reference electrode, [Cl-] is constant Potential of the cell only depends on [Fe2+] & [Fe3+] Unknown solution of [Fe2+] & [Fe3+] Pt wire is indicator electrode whose potential responds to [Fe2+]/[Fe3+] http:\\asadipour.kmu.ac.ir
B.) Reference Electrodes: (Instead of SHE) • Need one electrode of system to act as a reference against which potential • measurements can be made relative comparison. • Standard hydrogen electrodes are cumbersome • Requires H2 gas and freshly prepared Pt surface • Desired Characteristics: • a) known or fixed potential • b) constant response • c) insensitive to composition of solution under study • d) obeys Nernest Equation • e) reversible http:\\asadipour.kmu.ac.ir
Electrodes and Potentiometry • Reference Electrodes • 1.)Silver-Silver Chloride Reference Electrode Eo = +0.222 V Activity of Cl- not 1E(sat,KCl) = +0.197 V http:\\asadipour.kmu.ac.ir
Electrodes and Potentiometry • Reference Electrodes • 2.)Saturated Calomel Reference Electrode (S.C.E) • Saturated KCl maintains constant [Cl-] even with some evaporation Eo = +0.268 V Activity of Cl- not 1E(sat,KCl) = +0.241 V http:\\asadipour.kmu.ac.ir
Electrodes and Potentiometry • Indicator Electrodes • 1.)three Broad Classes of Indicator Electrodes • 1) Metal Electrodes • Develop an electric potential in response to a redox reaction at the metal surface • 2) Ion-selective (Membrane) Electrodes • Selectively bind one type of ion to a membrane to generate an electric potential • 3) Molecular Selective Electrode http:\\asadipour.kmu.ac.ir Remember an electric potential is generated by a separation of charge
1) Metallic Indicator Electrode (3 Main Types) a) Metallic Electrodes of the First Kind b) Metallic Electrodes of the Second Kind c) Metallic RedoxIndicators a) Metallic Electrodes of the First Kind i. Involves single reaction ii.Detection of cationederived from the metal used in the electrode iii.Example: use of copper electrode to detect Cu2+ in solution ½ reaction:Cu2+ + 2e-Cu (s) Eind gives direct measure of Cu2+: Eind= EoCu – (0.0592/2) log aCu(s)/aCu2+ since aCu(s) = 1: Eind = EoCu – (0.0592/2) log 1/aCu2+ or using pCu = -log aCu2+: Eind = EoCu – (0.0592/2) pCu http:\\asadipour.kmu.ac.ir
b) Metallic Electrodes of the Second Kind i. Detection of anion derived from the interaction with metal ion (Mn+) from the electrode ii.Anion forms precipitate or stable complex with metal ion (Mn+) iii.Example: Detection of Cl- with Ag electrode ½ reaction:AgCl(s) + e-Ag(s) + Cl- EO = 0.222 V Eind gives direct measure of Cl-: Eind = Eo – (0.0592/1) log aAg(s)aCl-/aAgCl(s) since aAg(s) and aAgCl(s)= 1 & Eo = 0.222 V: Eind = 0.222 – (0.0592/1) log aCl- http:\\asadipour.kmu.ac.ir
c) Metallic Redox Indicators i. Electrodes made from inert metals (Pt, Au, Pd) ii.Used to detect oxidation/reduction in solution iii. Electrode acts as e- source/sink iv.Example: Detection of Ce3+ with Pt electrode ½ reaction:Ce4+ + e-Ce3+ Eind responds to Ce4+: Eind = Eo – (0.0592/1) log aCe3+/aCe4+ http:\\asadipour.kmu.ac.ir
2) Membrane Indicator Electrodes a) General i. electrodes based on determination of cations or anions by the selective adsorptionof these ions to a membrane surface. ii.Often called Ion Selective Electrodes (ISE) or pIonElectrodes iii. Desired properties of ISE’s 1)minimal solubility – membrane will not dissolve in solution during measurement. – silica, polymers, low solubility inorganic compounds , (AgX) can be used 2)Need some electrical conductivity 3) Selectively binds ion of interest http:\\asadipour.kmu.ac.ir
Electrodes and Potentiometry • Indicator Electrodes • Ion-Selective Electrodes • Responds Selectively to one ion • Contains a thin membrane capable of only binding the desired ion • Does not involve a redox process Membrane contains a ligand (L) that specifically and tightly binds analyte of interest (C+) The counter-ions (R-,A-) can’t cross the membrane and/or have low solubility in membrane or analyte solution C+ diffuses across the membrane due to concentration gradient resulting in charge difference across membrane A difference in the concentration of C+ exists across the outer membrane. Potential across outer membrane depends on [C+] in analyte solution http:\\asadipour.kmu.ac.ir Remember an electric potential is generated by a separation of charge
Electrodes and Potentiometry • Indicator Electrodes • Ion-Selective Electrodes • Responds Selectively to one ion • Contains a thin membrane capable of only binding the desired ion • Does not involve a redox process C+ diffuses across the membrane due to concentration gradient resulting in charge difference across membrane A difference in the concentration of C+ exists across the inner membrane. Potential across inner membrane depends on [C+] in filling solution, which is a known constant Electrode potential is determined by the potential difference between the inner and outer membranes: where Einner is a constant and Eouter depends on the concentration of C+ in analyte solution http:\\asadipour.kmu.ac.ir Remember an electric potential is generated by a separation of charge
Electrodes and Potentiometry • Indicator Electrodes • Ion-Selective Electrodes • Responds Selectively to one ion • Contains a thin membrane capable of only binding the desired ion • Does not involve a redox process Electrode Potential is defined as: where [C+] is actually the activity of the analyte and n is the charge of the analyte http:\\asadipour.kmu.ac.ir
pH Electrode i. most common example of an ISE based on use of glass membrane that preferentially binds H+ ii.Typical pH electrode system is shown pH sensing element is glass tip of Ag/AgClelectrode Two reference electrodes here one SCE outside of membrane one Ag/AgCl inside membrane Combined electrod http:\\asadipour.kmu.ac.ir
Electrodes and Potentiometry • pH Electrodes • 1.)pH Measurement with a Glass Electrode Ag(s)|AgCl(s)|Cl-(aq)||H+(aq,outside) H+(aq,inside),Cl-(aq)|AgCl(s)|Ag(s) Outer reference electrode [H+] outside (analyte solution) [H+] inside Inner reference electrode Glass membrane Selectively binds H+ http:\\asadipour.kmu.ac.ir Electric potential is generated by [H+] difference across glass membrane
iii. pH is determined by formation of boundary potential across glass membrane Boundary potential difference (Eb) = E1 - E2 where from Nernst Equation: Eb = c – 0.592pH Selective binding of cation (H+) to glass membrane -log aH+ (on exterior of probe or in analyte solution) http:\\asadipour.kmu.ac.ir constant
Electrodes and Potentiometry • pH Electrodes • Glass Membrane • Irregular structure of silicate lattice Cations (Na+) bind oxygen in SiO4 structure http:\\asadipour.kmu.ac.ir
Electrodes and Potentiometry • pH Electrodes • Glass Membrane • Two surfaces of glass “swell” as they absorb water • Surfaces are in contact with [H+] http:\\asadipour.kmu.ac.ir
Electrodes and Potentiometry • pH Electrodes • Glass Membrane • H+ diffuse into glass membrane and replace Na+ in hydrated gel region • Ion-exchange equilibrium • Selective for H+ because H+ is only ion that binds significantly to the hydrated gel layer Charge is slowly carried by migration of Na+ across glass membrane Potential is determined by external [H+] http:\\asadipour.kmu.ac.ir Constant and b are measured when electrode is calibrated with solution of known pH
iii. pH is determined by formation of boundary potential across glass membrane At each membrane-solvent interface, a small local potential develops due to the preferential adsorption of H+ onto the glass surface. Glass Surface http:\\asadipour.kmu.ac.ir
Electrodes and Potentiometry • Junction Potential • 1.)Occurs Whenever Dissimilar Electrolyte Solutions are in Contact • Develops at solution interface (salt bridge) • Small potential (few millivolts) • Junction potential puts a fundamental limitation on the accuracy of direct potentiometric measurements • Don’t know contribution to the measured voltage Different ion mobility results in separation in charge Again, an electric potential is generated by a separation of charge http:\\asadipour.kmu.ac.ir
iv.Alkali Error ‚H+ not only cation that can bind to glass surface - H+ generally has the strongest binding ‚Get weak binding of Na+, K+, etc ‚Most significant when [H+] or aH+ is low (high pH) - usually pH $11-12 At low aH+ (high pH), amount of Na+ or K+ binding is significant increases the “apparent” amount of bound H+ http:\\asadipour.kmu.ac.ir
v.Acid Error ‚Errors at low pH (Acid error) can give readings that are too high ‚Exact cause not known - usually occurs at pH # 0.5 c) Glass Electrodes for Other Cations i. change composition of glass membrane ‚putting Al2O3 or B2O3 in glass ‚enhances binding for ions other than H+ ii. Used to make ISE’s for Na+, Li+, NH4+ http:\\asadipour.kmu.ac.ir
Example 18: The following cell was used for the determination of pCrO4: SCE||CrO42- (xM), Ag2CrO4 (sat’d)|Ag Calculate pCrO4 if the cell potential is -0.386. http:\\asadipour.kmu.ac.ir
16-1 The shape of a redox titration curve • A redox titration is based on an oxidation-reduction reaction between analyte and titrant. • Consider the titration of iron(II) with standard cerium(IV), monitored potentiometrically with Pt and calomel electrodes. The potentials show above is in 1 M HClO4 solution. Note that equilibria 16-2 and 16-3 are both established at the Pt electrode. http:\\asadipour.kmu.ac.ir
There are three distinct regions in the titration of iron(II) with standard cerium(IV), monitored potentiometrically with Pt and calomel electrodes. Before the equivalence point, where the potential at Pt is dominated by the analyte redox pair. At the equivalence point, where the potential at the indicator electrode is the average of their conditional potential. After the equivalence point, where the potential was determined by the titratant redox pair. http:\\asadipour.kmu.ac.ir
E of the cell Before the equivalence point: using analyte’s concentration to calculate E+ At the equivalence point: needs both redox pairs to calculate (why?) http:\\asadipour.kmu.ac.ir
After the equivalence point: Summary • The greater the difference in reduction potential between analyze and titrant, the sharper will be the end point. • The voltage at any point in this titration depends only on the ratio of reactants; it will be independent of dilution. • Prior to the equivalence point, the half-reaction involving analyze is used to find the voltage because the concentrations of both the oxidized and the reduced forms of analyte are known. • After the equivalence point, the half-reaction involving titrant is employed. At the equivalence point, both half-reactions are used simultaneously to find the voltage. There are two special points during the above titration process: (1) when V = ½ Ve, [Fe3+] = [Fe2+] and E+ = E°(Fe3+ | Fe2+) ; (2) when V = 2 Ve, [Ce4+] = [Ce3+] and E+ = E°(Ce4+ | Ce3+) = 1.70 V. http:\\asadipour.kmu.ac.ir
Titration curve.xlsx http:\\asadipour.kmu.ac.ir