parallel bonds
E N D
Presentation Transcript
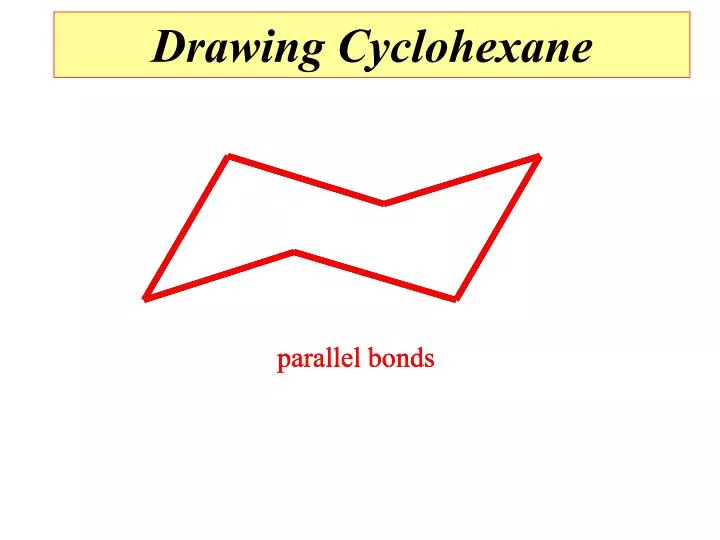
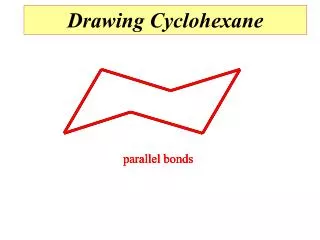
parallel bonds parallel bonds parallel bonds Drawing Cyclohexane
equatorial bonds axial bonds
4 6 5 1 3 2 Numbering Cyclohexane Start anywhere and number consecutive carbons.
Tetrahedral Carbons in Cyclohexane 1 tetrahedral carbon
2 tetrahedral carbon
3 tetrahedral carbon
4 tetrahedral carbon
5 tetrahedral carbon
6 tetrahedral carbon
4 6 5 1 3 2 6 axial bonds (green) (3 straight up; 3 straight down) 6 equatorial bonds (blue) (3 slightly up; 3 slightly down)
Disubstituted Isomers of Cyclohexane 1 2 two equatorial substituents (purple) name of cpd: trans-1,2-di . . . cyclohexane and . . .
1 2 two axial substituents (purple) name of cpd: trans-1,2-di . . . cyclohexane are conformational isomers of one another. Also, . . .
1 3 two equatorial substituents on carbons 1 & 3 name of cpd: cis-1,3-di . . . cyclohexane and . . .
1 3 two axial substituents on carbons 1 & 3 name of cpd: cis-1,3-di . . . cyclohexane) are conformational isomers of one another. Furthermore, . . .
4 1 two equatorial substituents on carbons 1 & 4 name of cpd: trans-1,4-di . . . cyclohexane) and . . .
1 4 two axial substituents on carbons 1 & 4 name of cpd: trans-1,4-di . . . cyclohexane are conformational isomers of one another.
the other chair conformation hold red bonds in a plane flip this C above the plane flip the other C below the plane Chair Conformations of Cyclohexane you get
number carbons in this conformation 6 4 5 2 3 1 5 number the same carbons in this conformation 6 1 3 4 2
6 4 5 C4 is above the plane, while... 2 3 1 C4 is below the plane. 5 6 C1 is below the plane and ... 1 3 4 C1 is above the plane and... 2 given the plane formed by carbons 2, 3, 5, and 6
6 4 equatorial bonds here. 5 axial bonds here become ... 2 3 1 5 1 axial bonds here, and... 6 equatorial bonds here become ... 3 4 2
4 6 5 1 3 2 above below below below above above Equatorial bonds alternate being above and below the axial bonds on a given carbon atom and . . .
4 6 5 1 3 2 above above above below below below axial bonds alternate being above and below the equatorial bonds on a given carbon atom.
The End And what a nifty cool story it was . . .