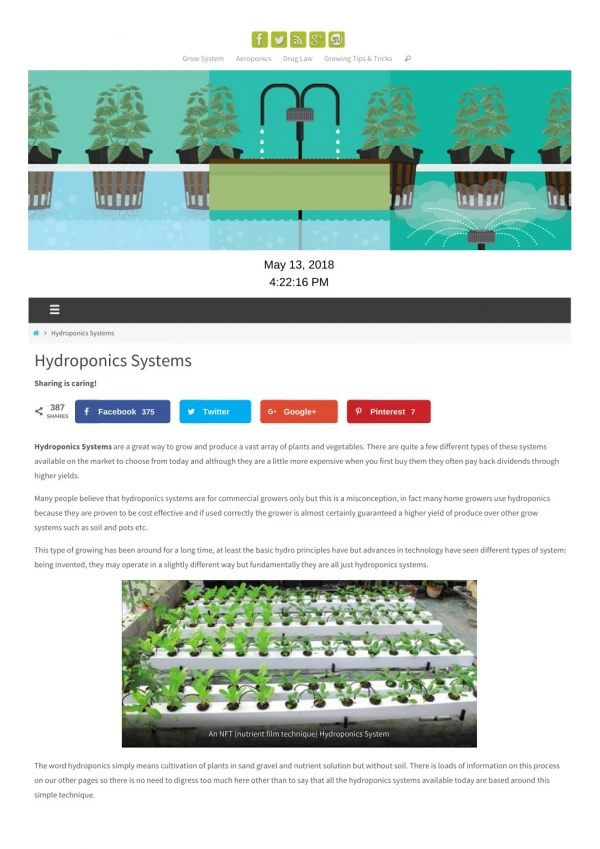
HYDROPONICS
HYDROPONICS. Nutrient Quality and Applications Stuart LaPlace Lecturer : CFB College Consultant: LLM & Co. Ltd. How do plants acquire these nutrients?.

HYDROPONICS
E N D
Presentation Transcript
HYDROPONICS Nutrient Quality and Applications Stuart LaPlace Lecturer : CFB College Consultant: LLM & Co. Ltd
How do plants acquire these nutrients? • Upon exposure to sunlight, plants transpire (loose water) via their leaves in a process called transpiration. Once these nutrients are dissolved in water (hydration shell) they are taken up along with the water as part of the transpiration process. Most of this water is taken along the Apoplast route (from cell wall to cell wall) while the balance is taken alone the Symplast route (through the cytoplasm)
How are these nutrients organized? • Upon entry into the cytoplasm the nutrients are organized into groups by presence of the element Potassium (K) to make specific proteins. For example Nitrogen ions are grouped with a single Magnesium (Mg) ion to make the protein chlorophyll.
Adding your nutrient formulae • You should try your very best to use dissolved nutrients for your hydroponic formulae. The most ideal combination will be a 3 part combination. That is a mix that has your primary nutrients (Nitrogen Phosphorus and Potassium), secondary nutrients (Mg, Ca, S) and your trace elements (Zn, Co, Mn). • It is important that these elements be kept separate and apart even when you are adding them to your reservoir. That is the primary should be added first, allowed to circulate for a few minutes before you add the other combinations to avoid nutrient lock.
What is nutrient lock, how does it occur and how can it be avoided? Nutrient lock is a process by which nutrients become unavailable to a plant. This happens when elements combine to form compounds which become inaccessible to the plant. There are a number of ways this can happen : • When nutrients are mixed inappropriately in a hydroponic reservoir some elements displace others easily and form compounds which are not easily broken down. • If you don’t have enough water to nutrient ratio
Nutrient lock • If you do not top off regularly to compensate for evaporation and water loss by transpiration. • Some elements are higher in the activity series compared to others and in excess will displace others and form compounds which will cause nutrient deficiency even though it may be present in solution.
Nutrient Lock • To avoid these problem requires due diligence in understanding the crop grown as well as your water input versus the rate of water loss. For instance a 60 plant hydroponic system under 50% shade where plants are in bloom stage in a closed system will lose 1-2 gallon of water in 48 hrs. This means in order to keep your nutrients in solution and prevent nutrient lock you have to top off (return to original level).
When do you stop topping off? • As a rule of thumb when trying to utilize your nutrients efficiently, discard the contents of the reservoir when the water you have added over the period equals the amount you started out with initially.
What is pH and how important is it to my plants? • pH is a measure of hydrogen ions in a solution. This is extremely important to plants because plants can die quickly if the pH is too high (alkaline) or too low (acidic). Crops using take up nutrients rapidly when the ph is slightly acidic. This can also be attributed to the quantity of cations (+ ions) as compared to the anions (-Ions).
What causes my pH to change and how can I correct it? • When a plant takes up one positive element from your hydroponic solution it must release one H+ ion into the same solution. The more positive elements it takes up from solution the more H+ ions it releases to the solution. As mentioned before pH is a measure of H+ ions in solution and therefore the more released in solution the lower or more acidic the ph becomes. The solution will be to dilute frequently.
What is TDS, EC and how are they used to govern my hydroponics system? TDS : Total dissolved solids • This represents the amount of dissolved particles and is measured in ppm • A solution reading 1200ppm means that for every million molecules in solution 1200 of them are dissolved ions. • Not very reliable for use on hydroponics systems as various scales exist across instruments.
Electric Conductivity • EC or electric conductivity of a solution is a measure of the ability for that solution to conduct electricity. • Measured in micro-Siemens (µs) • Very effective and most suitable in hydroponic system applications and provides a set standard world wide. • ppm = ec x 0.67
There are many factors that can affect the E.C. level in your reservoir and growing medium • The temperature of the nutrient solution can affect the ability of the nutrient tester to read accurately. • Evaporation not only causes the concentration of the solution to increase, but it will cause nutrient to be left behind and build up
Hydroponic Facts • E.C. and P.P.M. Meters cannot tell the gardener exactly which elemental salts or metals are low or which are too high. • some ingredients that will not register on nutrient testers because they do not conduct electricity. Example chelates • Nutrients that are separated into an A part and a B part will not contain anti-binding agents.
Facts cont’d • Clearing solutions can be applied to plants if the grower suspects a nutrient toxicity. • Clearing solutions latch on to salts in the plants as well as in the growing medium and leech the salts out • Having a clearing solution on hand, could save a crop from toxicity when it seems to be doomed. • The amount of nutrient in the solution that is fed to a plant is as important as the quality of the nutrient.
Sodium (Na+) • Partly replaces Potassium in its role when potassium becomes deficient in some plants. • Helps plants over come plant stress • Boron (B) • Boron is also involved in the carbohydrate metabolism in plants, protein synthesis, seed and cell wall formation, germination of pollen grains and growth of pollen tubes and sugar translocation
Calcium (Ca 2+) • Ca2+ ions are an essential component of plant cell walls and cell membranes, and are used as cations to balance organic anions in the plant vacuole. • Calcium is needed to form the pectin in the middle lamella of newly formed cells. • Without calcium, the cell walls are unable to stabilize and hold their contents. This is particularly important in developing fruits. Without calcium, the cell walls are weak and unable to hold the contents of the fruit.
Sulphur (S) • Sulphur is a structural component of some amino acids and vitamins, and is essential in the manufacturing of chloroplasts • Sulphur is also found in the Iron Sulphur complexes of the electron transport chains in photosynthesis. • It is immobile and deficiency therefore affects younger tissues first. • Symptoms of deficiency include yellowing of leaves and stunted growth
Cobalt (Co) • Cobalt delays ageing and is used for keeping leaves fresh and fruits fresh, • Biocidal and Antifungal Activity • Cobalt acts mainly through arresting the conversion of methionine to ethylene Manganese (Mn) • Manganese is necessary for building the chloroplasts. • Manganese deficiency may result in coloration abnormalities, such as discoloured spots on the foliage
Copper (Cu2+) • It functions as a catalyst in photosynthesis and respiration. • Copper is important in carbohydrate and protein metabolism. • It is important to the formation of lignin in plant cell walls which contributes to the structural strength of the cells, and the plant. • Copper also affects the flavor, the storageability, and the sugar content of fruits.
Copper cont’d • Cu: Zn Balance: High Zn levels will reduce Cu availability. • Cu: N Balance: High N uptake in the presence of marginal Cu levels can lead to a reduction of Cu transport into the growing tips of plants. • Root Growth: Copper is the most immobile micronutrient, therefore anything that inhibits new root growth will inhibit Cu uptake.
Iron (Fe2+) • Iron is necessary for photosynthesis and is present as an enzyme cofactor in plants. • Iron deficiency can result in interveinal chlorosis and necrosis.
Nutrient availability across systems Season peppers in Hydroponics Season pepper in Organoponics
Potassium (K+) • The main role of potassium is to provide the appropriate ionic environment for metabolic processes in the cytosol, and as such functions as a regulator of various processes including growth regulation • Plants require potassium ions (K+) for protein synthesis and for the opening and closing of stomata • Typical symptoms of potassium deficiency in plants include brown scorching and curling of leaf tips as well as (yellowing) between leaf veins. Purple spots may also appear on the leaf undersides
Zinc (Zn2+) • Zinc is required in a large number of enzymes and plays an essential role in DNA transcription. • A typical symptom of zinc deficiency is the stunted growth of leaves, commonly known as "little leaf" and is caused by the oxidative degradation of the growth hormone auxin Nitrogen (N) • Nitrogen is an essential component of all proteins. • Nitrogen deficiency most often results in stunted growth, slow growth, and chlorosis. • Nitrogen deficient plants will also exhibit a purple appearance on the stems, petioles and underside of leaves
Shade dictates nutrient uptake Lettuce in direct sunlight at 23 days Lettuce in 60% shade at 23 days
Phosphorus (P) • Phosphorus is important in plant bioenergetics. As a component of ATP, phosphorus is needed for the conversion of light energy to chemical energy (ATP) during photosynthesis. • phosphorus is important for plant growth and flower/seed formation. • High phosphorus deficiency causes necrosis or death of leaves. Magnesium (Mg 2+) • Magnesium is part of the chlorophyll in all green plants and essential for photosynthesis. • Without magnesium plants would die because it would not be able to photosynthesize. So it would not have any food which is glucose as energy for growth and repair. The magnesium is needed so that the chlorophyll in the chloroplasts can capture the sun's light energy and use it for photosynthesis.
Excessive Magnesium uptake Lettuce under direct sunlight Lettuce under 60% shade
Nutrient management Poor nutrient quality Rich nutrient quality
Molybdenum (Mb) • Molybdenum is a cofactor to enzymes important in building amino acids. Nickel (Ni) • Nickel is essential for activation of urease, an enzyme involved with nitrogen metabolism that is required to process urea. • Without Nickel, toxic levels of urea accumulate, leading to the formation of necrotic lesions. Chloride (Cl-) • Chlorine is necessary for osmosis and ionic balance; it also plays a role in photosynthesis.
The End Thank you