Junk DNA and DNA editing
220 likes | 344 Vues
This discussion explores the complexities of junk DNA and its roles in the genome. While protein-coding genes constitute only about 2% of human DNA, non-coding elements play vital functions, including gene regulation and structural integrity. Retrotransposons, significant mobile DNA elements, comprise nearly 40% of our genome, driving both mutation and genetic innovation. We delve into the mechanisms of DNA editing and its potential in combating retroviral replication, emphasizing the balance between genomic complexity and stability.

Junk DNA and DNA editing
E N D
Presentation Transcript
Junk DNA and DNA editing Shai Carmi Bar-Ilan, BU מוצ"ש י"ג אייר 17/05/2008
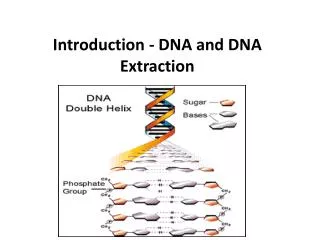
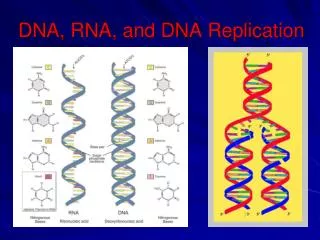
Genome structure • DNA has mostly evolved to store the code of the proteins its host cell is using. • Thus, the main functional units of any genome are protein coding genes. • The central dogma of molecular biology: DNA→RNA → Protein 5’ 3’ A,C,G,T A,C,G,U Final product: Proteins are the cellular machinery 20 amino acids
מותר האדם מן הבהמה אין? • In human, protein coding sequences are only 2% of the genome. • All animals have the same order of magnitude of genes (few tens of thousands). • Does non-coding DNA determines complexity? • Is everything else junk?
Non-coding DNA • The rest codes for introns, promoters and enhancers (regulation of expression), structural sequences (e.g. telomeres), non-coding RNAs such as rRNA and tRNA (translation), micro-RNA (silencing), snRNA (splicing). • But this is not all! • Almost HALF of the human genome is made of mobile elements. • Pieces of ~100-10k base pairs moving around the genome in a cut&paste or copy&paste mechanisms.
DNA transposons • DNA transposons: cut&paste using the enzyme transposase(3% of the genome). • Sometimes transfers also host sequences. • Increases the genome volume only through repeats at the edgesor if happens during S-phase.
Retrotransposons • Retrotransposons: copy&paste mechanism through RNA intermediate. • Main classes: • LTR (retrovirus like, 8.7% of the genome). • LINE (Long interspersed nuclear elements, 21.3%). • SINE (Short interspersed nuclear elements, 13.6%). • Retrotransposons behave like retroviruses. • What are retroviruses?
Retroviruses • Retroviruses are pieces of (ss) RNA (DNA in other viruses) wrapped in a capsid and envelope. • They penetrate into the cell, and use the cell machinery to replicate, assemble a new virus, and infect another cell. • Example: HIV. Few thousand bases
Retroviral proteins (advanced) • Pol: Encodes a polyprotein with- • protease (cleavage of the retrovirus proteins). • Reverse transcriptase (copy the RNA to DNA). • RnaseH (degradation of RNA after reverse transcription). • Integrase (integration of the DNA into the genome). • Gag: Codes for core and structural proteins of the virus. • Env: Glycoprotein that recognizes membrane receptors of the host cell and initiate the process of infection. • Complex splicing pattern, with partial overlap and frameshifting
Retrotransposons • It is commonly believed that ancient retroviral infection in the germ line is the origin of nowadays retrotransposons. • How did they occupy 40% of the genome? • Transcription: genomic DNA→RNA. • Translation of viral proteins (if possible). • Reverse transcription: RNA → DNA by reverse transcriptase. • Insertion into new genomic locations, increasing the number of genomic copies of the sequence. • Mobile elements are like double edge sword. RETRO: violating the central dogma!
Why are retrotransposons good? • Serve as reservoir of sequences for genetic innovation. • Retroviral proteins have DNA binding capabilities which can be exploited by the host cell. • Regulate expression levels of existing genes. • Change gene regulation networks: • By copying a promoter, two sequences are controlled by the same transcription factors (or in other cases by RNA binding proteins or miRNA).
Why are retrotransposons bad? • Retroelements generate mutations, through direct insertion into genes, or unequal homologous recombination. • Responsible to 0.3-0.5% of all genetic disorders (e.g. hemophilia). • Change the normal transcription of the gene (alter promoter activity, anti-sense transcription, silencing via methylation or miRNA binding). • Alternative splicing and protein isoforms.
Examples How can we stop them ???
Inhibition of retroelements Few mechanisms exist: • Accumulation of mutations results in non-autonomous elements. • Methylation and heterochromatin formation attenuates transcription (LINE). • RNA interference. • DNA editing(more to come). • Did we succeed? • Probably we did: • Here we are, more complex than any other organism. • Most elements are inactive–only Alu and L1 are active with insertion once in 100 births.
Basics of DNA editing • The APOBEC3 family of proteins was found to restrict retroviral replication. One of its mechanisms of operation is by “Cytosine Deamination of the (-) strand DNA strand after reverse transcription”. Meaning… • APOBEC catalyzes some chemical modification of the DNA just before it is integrated into the genome, eventually generating G→A mutation (editing). • (localization varies nucleus/cytoplasm). • Inducing tens/hundreds of mutations (uracil excision?). • Editing itself is not sufficient to stop replication- other mechanisms are also used. Useful in the immune system to generate new antibodies!
Evolution of APOBEC • APOBEC3G is one of the most positively selected genes (=changes the fastest). • Ongoing arms race with HIV. • In response to APOBEC, HIV developed the Vif protein that can ubiquitinate APOBEC (=send it to “recycle” (proteasome)). • Different APOBECs restrict retroviruses/transposons in different mechanisms (e.g., binding to RNA and blocking reverse transcription).
DNA editing in the genome • Some retrotransposons were edited by APOBEC, but yet integrated into the genome. • New mechanism of mutagenesis. • So far, almost neglected by geneticists. • Together with Erez Levanon, HMS. • Analyzed retroelements in mouse, human and chimp,applying new statistical approach.
Main results • Editing has fingerprints in thousands of mouse IAP/MusDretroelements, with distinguished motifs. • Predicting hundreds of thousands editing sites. • Edited IAPs are transcribed more than non-edited. • Some edited IAPs overlap with introns and exons. • Phylogenetic tree can be changed if considering editing information. • Editing also in non-LTR, LINE mouse elements. • Editing in human and chimp HERV retroelements.
DNA editing demonstration • Comparing two mouse IAPs. • chr9:114987516-114993954chr8:28575443-28581824 • One cluster of 68 consecutive G→A! • Total 176/202 G→A mismatches. Can editing accelerate evolution? Easily available raw material for the generation of new functions! (for example: any editing in TGG creates premature stop codon).
DNA editing phylogenetics Same tree, masking the editing. Automatically generated genetic tree. If two sequences are the same except for G→A mutation, the sequence with ‘G’ must precede the one with ‘A’. Thus we can build the tree of elements. Editing affects phylogenetics!
Summary • Significant fraction of the DNA originates from infection by ancient RNA viruses, spreading through the genome by reverse transcription and replication. • Some of them ‘domesticated’ to benefit the host cell (not really junk!), but some induce deleterious mutations. • One of the mechanisms to restrict retrotransposition is editing them before integration into the genome. • Many genomic sites are ‘edited’ due to this restriction activity. • New mechanism of mutagenesis, potentially leading to evolution of new molecules or function (for example, HIV drug resistance).