Chapter 28 Atomic Physics
Chapter 28 Atomic Physics. The Hydrogen Atom The Bohr Model Electron Waves in the Atom.

Chapter 28 Atomic Physics
E N D
Presentation Transcript
Chapter 28 Atomic Physics The Hydrogen Atom The Bohr Model Electron Waves in the Atom
Wave Function, yThe value of 2 for a particular object at a certain place and time is proportional to the probability of finding the object at that place at that time.The Heisenberg Uncertainty Principle Momentum and position DxDp ≥ h/4p Energy and time DEDt ≥ h/4p
Rutherford Model Predicts: • A continuous range of frequencies of light emitted • Unstable atoms • These are inconsistent with experimental observations Why ?
Niels Henrik David Bohr 1885-1962
Quantized orbits Each orbit has a different energy
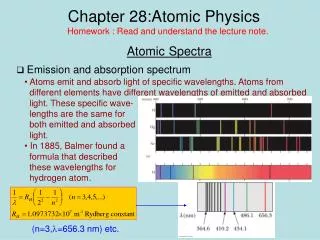
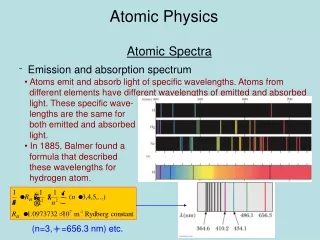
1/l=R(1/22-1/n2), n=3,4,… for Balmer series where Rydberg constant R=1.097x107 m-1
Equations Associated with The Bohr Model Electron’s angular momentum L=Iw=mvrn=nh/2p, n=1,2,3 n is called quantum number of the orbit Radius of a circular orbit rn=n2h2/4p2mkZe2=(n2/Z)r1 where r1=h2/4p2mke2=5.29x10-11 m (n=1) r1 is called Bohr radius, the smallest orbit in H Total energy for an electron in the nth orbit: En=(-2p2Z2e4mk2/h2)(1/n2)=(Z2/n2)E1 where E1=-2p2Z2e4mk2/h2 =-13.6 eV (n=1) E1 is called Ground State of the hydrogen Both orbits and energies depend on n, the quantum number
To break a hydrogen atom apart requires 13.6 eV + 13.6 eV = + electron Proton Hydrogen atom e v=2.2x106 m/s p Electron orbit r=0.053 nm
Question: In the Bohr model of the hydrogen atom, the electron revolves around the nucleus in order to • emit spectral lines • produce X rays • form energy levels that depend on its speed • keep from falling into the nucleus Answer: d
Question: A hydrogen atom is in its ground state when its orbital electron • is within the nucleus • has escaped from the atom • is in its lowest energy level • is stationary Answer: c
Which one of the following statements is the assumption that Niels Bohr made about the angular momentum of the electron in the hydrogen atom? (a) The angular momentum of the electron is zero. (b) The angular momentum can assume only certain discrete values. (c) Angular momentum is not quantized. (d) The angular momentum can assume any value greater than zero because it’s proportional to the radius of the orbit. (e) The angular momentum is independent of the mass of the electron. X
Example: Find the orbital radius and energy of an electron in a hydrogen atom characterized by principal quantum number n=2. Solution: For n=2, r2=r1n2=0.0529nm(2)2=0.212 nm and E2=E1/n2=-13.6/22 eV=-3.40 eV
1.The kinetic energy of the ground state electron in hydrogen is +13.6 eV. What is its potential energy? (a) –13.6 eV (b) –27.2 eV (c) zero eV (d) +27.2 eV (e) +56.2 eV 2. An electron is in the ground state of a hydrogen atom. A photon is absorbed by the atom and the electron is excited to the n = 2 state. What is the energy in eV of the photon? (a) 13.6 eV (b) 3.40 eV (c) 0.54 eV (d) 10.2 eV (e) 1.51 eV X X
Line and Absorption Spectra hf=Eu- El hc/l=Eu - El 1/l=(1/hc)Eu- El 1/l=(2p2Z2e4mk2/ch3)(1/nl2-1/nu2)
Each atom in the periodic table has a unique set of spectral lines. Which one of the following statements is the best explanation for this observation? (a) Each atom has a dense central nucleus. (b) The electrons in atoms orbit the nucleus. (c) Each atom has a unique set of energy levels. (d) The electrons in atoms are in constant motion. (e) Each atom is composed of positive and negative charges. X
Complete the following statement: An individual copper atom emits electromagnetic radiation with wavelengths that are (a) evenly spaced across the spectrum. (b) unique to that particular copper atom. (c) the same as other elements in the same column of the periodic table. (d) unique to all copper atoms. (e) the same as those of all elements. X
Wave-Particle Duality l = h/p= h/mv
The quantized orbits and energy states in the Bohr model are due to the wave nature of the electron, and the electron wave functions can only occur in the form of standing waves. Implication: The wave-particle duality is at the root of atomic structure
Condition for orbit An electron can circle an atomic nucleus only if its orbit is a whole number of electron wavelengths in circumference Condition for orbit stability nl=2prn, n=1,2,3…
Unless a whole number of wavelengths fits into the wire loop, destructive interference causes the vibrations to die out rapidly n=4
de Broglie wavelength is l=h/mv and the speed of the electron in a hydrogen is v=2.2x106 m/s so l=h/mv =6.63x10-34Js/(9.1x10-31kg)(2.2x106m/s) =3.3x10-10 m 2pr1=2px5.29x10-11m=3.3x10-10 m The orbit of the electron in a hydrogen atom corresponds to one complete electron wave joined on itself!
Question: With increasing quantum number, the energy difference between adjacent energy levels • decreases • remains the same • increases • sometimes decreases and sometimes increases Answer: a
Question: An atom emits a photon when one of its electrons • collides with another of its electrons • is removed from the atom • undergoes a transition to a quantum state of lower energy • undergoes a transition to a quantum state of higher energy Answer: c
Question: The bright-line spectrum produced by the excited atoms of an element contains wavelength that • are the same for all elements • are characteristic of the particular element • are evenly distributed throughout the entire visible spectrum • are different from the wavelength in its dark-line spectrum Answer: b
Question: According to the Bohr model, an electron can revolve around the nucleus of a hydrogen indefinitely if its orbit is • a perfect circle • sufficient far from the nucleus to avoid capture • less than one de Broglie wavelength in circumference • exactly one de Broglie wavelength in circumference Answer: d
Question: How can the spectrum of hydrogen contains so many lines when hydrogen contains only one electron? Answer: The electron in the hydrogen atom can be in any of a nearly infinite number of quantized energy levels. A spectral line is emitted when the electron makes a transition from one discrete energy level to another discrete energy of lower energy. A collection of many hydrogen atoms with electrons in different energy levels will give a large number of spectral lines.
Example: An electron collides with a hydrogen atom initially in its ground state and excites it to a state of n=3, How much energy was transferred to the hydrogen atom in this inelastic (KE not conserved) collision? Solution:DE=E1(1/n2f-1/n2i) Here ni=1, nf=3 and E1=13.6 eV DE=E1(1/n2f-1/n2i)=E1(1/32-1/12)=-13.6(-8/9)eV =12.1 eV
The presence of definite energy levels in an atom is true for all atoms. Quantization is characteristic of many quantities in nature • Bohr’s theory worked well for hydrogen and for one-electron ions. But it did not prove as successful for multielectrons. • It is quantum mechanics that finally solved the problems
Early Quantum Theory • Quantum energy: E=hf • Photoelectric effect: hf=KEmax+Wo • De Broglie wavelength: l=h/mv • Bohr theory: L=mvr=nh/2p En=E1/n2 where E1=-13.6 eV • Wave-particle duality
Limitations of the Bohr Theory • Unable to predict the line spectra for more complex atoms • Unable to predict the brightness of spectral lines of hydrogen • Unable to explain the fine structure • Unable to explain bonding of atoms in molecules, solids and liquids • Unable to really resolve the wave-particle duality
Quantum Mechanics • It solves all these problems and has explained a wide range of physical phenomena. • It works on all scales of size. Classical physics is an approximation of quantum physics • It uses an abstract mathematical formulation dealing with probabilities
Quantum mechanics Bohr Model No precise orbits of electrons, only the probability of finding a given electron at a given point Definite circular orbits of electrons
Upon which one of the following parameters does the energy of a photon depend? (a) mass (c) polarization (e) phase relationships (b) amplitude (d) frequency For which one of the following problems did Max Planck make contributions that eventually led to the development of the “quantum” hypothesis? (a) photoelectric effect (d) the motion of the earth in the ether (b) uncertainty principle (e) the invariance of the speed of light through vacuum (c) blackbody radiation curves X X
Description of waves Type of waves Variable physical quantity Water waves Height of the water surface Sound waves Pressure in the medium Light waves Electric and magnetic fields Matter waves Wave function, • , the amplitude of a matter wave, is a function of time and position
Probability Density 2 The value of 2 for a particular object at a certain place and time is proportional to the probability of finding the object at that place at that time. For example: 2 =1: the object is definitely there 2 =0: the object is definitely not there 2 =0.4: there is 40% chance of finding the object there at that time. 2 starts from Schrodinger’s equation, a differential equation that is central to quantum mechanics
Why 2? Why not ? • Amplitude of every wave varies from –A to +A to –A to +A and so on (A is the maximum absolute value whatever the wave variable is). • A negative probability is meaningless. • 2 gives a positive quantity that can be compared with experiments.
The key point to the wave function is that the position of a particle is only expressed as a likelihood or probability until a measurement is made.
The probability the electron will be found at the particular position is determined by the wave function illustrated to the right of the aperture. When the electron is detected at A, the wave function instantaneously collapses so that it is zero at B.
Example: Compare the de Broglie wavelength of 54-eV electrons with that of a 1500-kg car whose speed is 30 m/s. Solution: For the 54-eV electron: KE=(54eV)(1.6x10-19J/eV)=8.6x10-18 J KE=1/2 mv2, mv=(2mKE)1/2 l=h/mv=h/(2mKE)1/2= 1.7x10-10 m The wavelength of the electron is comparable to atomic scales (e.g., Bohr radius=5.29x10-11 m). The wave aspects of matter are very significant. For the car: l=h/mv=6.63x10-34 J•s/(1.5x103)(30m/s)= 1.5x10-38 m The wavelength is so small compared to the car’s dimension that no wave behavior is to be expected.