Atomic Physics
Atomic Physics. Chapter 28. Atomic Models. Introduction. How do neon signs work?. Our main focus will be on the hydrogen atom. It is the simplest atomic system. Why is it important to study the hydrogen atom?


Atomic Physics
E N D
Presentation Transcript
Atomic Physics • Chapter 28
Introduction • How do neon signs work?
Our main focus will be on the hydrogen atom. • It is the simplestatomic system.
Why is it important to study the hydrogen atom? • Studying the quantum numbers for the allowed states of hydrogen will help us to describe the allowed states of more complex atoms. • The hydrogen atom is an ideal system for relating theory to experimentation. • Much that we learn about hydrogen can be extended to single electron ions like He and Li.
Early Models Of The Atom • The Greek model • Tiny, hard, indestructible sphere 3
The J. J. Thomson model • A volume of positive charge is embedded with negative charges called “electrons”
The Rutherford model • A positive nucleus orbited by electrons. • The nucleus contains 99.9% of the atom’s mass
The Rutherfordmodel • Which force holds the electrons in orbit? • The Coulomb force
Problems with the Rutherford Model • There were two basic difficulties with the Rutherford model. • It could not explain why atoms radiate discrete frequencies. • Accelerating electrons should radiate electromagnetic waves.
Electron Transitions • Using a high voltage to move electrons through a gas causes the gas electrons to become excited and to jump from lower energy levels to higher energy levels. • Photons of various wavelengths are produced when electrons fall from higher energy levels to lower energy levels.
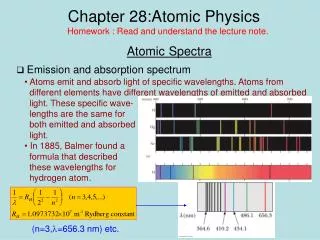
Emission Spectra • The emission spectrum of hydrogen • Can be produced by applying a high voltage across an evacuated glass tube filled with hydrogen • The observed wavelengths are characteristic only of hydrogen 279, 57
The Balmer Series • In the Balmer Series • nf= 2 • There are four prominent wavelengths • 656.3 nm(red) • 486.1 nm(green) • 434.1 nm(purple) • 410.2 nm(deep violet) 278, 28.7
The Balmer Series Wavelength Equation • RH is the Rydberg constant RH = 1.0973732 x 107m-1
Two Other Important Series • Lyman series(UV) • nf= 1 • Paschen series(IR) • nf= 3 70
Spectral Lines • How many different spectral lines could be produced by an electron in the n = 3 state? Three
How many different spectral lines could be produced by an electron in the n = 4 state? Six
Photon Energy • The equation for determining the energy of the emitted photon in any series:
The Absorption Spectrum • An element can absorb the same wavelengths that it emits. • The spectrum consists of a series of dark lines.
Identifying Elements • The absorption spectrumwas used to identify elements in the solar atmosphere were identified in this way. • Helium was discovered.
Thermal vs. Atomic Spectra • How could you tell if the light from a candle flame is thermal or atomic in origin?
Auroras • What is the origin of the colors in the aurora borealis?
High speed particles from space interact with the earth’s magnetic field.
The Bohr Theory Of Hydrogen • At the beginning of the 20th century, scientists wondered why atoms only radiated certain wavelengths. • Bohr provided an explanation.
Four Assumptions of The Bohr Theory • 1) The electron orbits the proton due to the Coulomb force which produces centripetal acceleration.
2) Only certain electron orbits are stable and do not radiate energy.
3) Radiation is only emitted when an electron drops from a more energetic state to a lower state.
4) The radius of the electron’s orbit is determined by the electron’s orbital angular momentum. 28.6
Total Energy of the Hydrogen Atom • The total energy of the hydrogen atom can be determined by using this equation.
The Bohr Radius • An electron can exist only in certain allowed orbits determined by the integer n. • When n = 1, we have what is known as the Bohr radius (ao). ao = 0.0529 nm
Orbital Radii • A general equation for finding the radius of any orbit:
Energy States • The energy for various energy states can be found by using: n= 1 is the ground state
Ionization Energy • The minimum energy required to ionize the atom is called the ionization energy. • An electron is completely removed from the atom.
The Hydrogen Spectrum • The general expression for determining wavelengths of the various series in the hydrogen spectrum
Bohr’s Correspondence Principle • Quantum mechanics is in agreement with classical physics when the energy differences between quantized levels are very small.
Successes of the Bohr Theory • It accounted for theBalmer seriesand other series.
It predicted a value for theRydberg constantthat agreed strongly with the experimental value.
It also works with hydrogen-like (one electron) atoms. • Singly ionized helium
It also works with hydrogen-like (one electron) atoms. • Doubly ionized lithium
It also works with hydrogen-like (one electron) atoms. • Triply ionized beryllium
Four Quantum Numbers • The state of an electron is specified by four quantum numbers. • These numbers describe all possible electron states. • The total number of electrons in a particular energy level is given by:
Principle Quantum Number • The principal quantum number(n) wheren = 1, 2, 3, … • Determines the energy of the allowed states of hydrogen • States with the same principal quantum number are said to form a shell • K, L, M, … (n = 1, 2, 3, …)
Orbital Quantum Number • The orbital quantum number(l)where l ranges from 0 to (n – 1) in integral steps • Allows multiple orbits within the same energy level • Determines the shape of the orbits • States with given values of n and lare called subshells • s(l = 0),p(l = 1),d(l = 2),f(l = 3), etc…