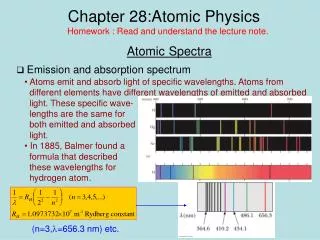
Atomic Physics
Learn about calculating kinetic energy of electrons accelerated through a potential difference, determining X-ray wavelengths, working with Planck's constant, and solving photoelectric effect equations.

Atomic Physics
E N D
Presentation Transcript
Calculate the Kinetic Energy gained by an electron when it is accelerated through a P.D. of 50kV in an X-ray tube • Firstly we know that KE is measured in Joules. • JDeV….. • eV = ½mv2(KE) • eV = (1.6 X 10-19)(50000) = 8 X 10-15 Joules = KE
Calculate the minimum wavelength of an X-ray emitted from the anode.(Planck’s = 6.6 X 10-34Js; c = 3 X 10-8ms-1; Charge on electron = 1.6 X 10-19C) • What formulae do we know with Planck? • E = hf • E = hc/λ • We know E from last question = 8 X 10-15 J
Calculate the minimum wavelength of an X-ray emitted from the anode.(Planck’s = 6.6 X 10-34Js; c = 3 X 10-8ms-1; Charge on electron = 1.6 X 10-19C) • E = hc/λ • 8 X 10-15 J = (6.6 X 10-34)(3 X 10-8ms-1)/λ • λ = (6.6 X 10-34)(3 X 10-8ms-1)/ 8 X 10-15 • λ = 2.475 X 10-27 m
The work function of zinc is 6.9 X 10-19J. What is the minimum frequency of UV radiation that will cause the photoelectric effect to occur in zinc. (Planck’s = 6.6 X 10-34Js) • The main equation that contains work function (and the most important is E = Φ + ½ mv2 • This is also written as: • hf = hfo + ½ mv2
The work function of zinc is 6.9 X 10-19J. What is the minimum frequency of UV radiation that will cause the photoelectric effect to occur in zinc. (Planck’s = 6.6 X 10-34Js) • E = Φ + ½ mv2 • hf = hfo + ½ mv2 • Hence Φ = hfo • fo = Φ/h = 6.9 X 10-19J/ 6.6 X 10-34Js • fo = 1.05 X 10-15 Hz
Zinc is illuminated with UV light of wavelength 240nm. The work function is 4.3 eV. Calculate the threshold frequency of zinc and Max KE of emitted electrons.(Planck’s = 6.6 X 10-34Js; c = 3 X 10-8ms-1; Charge on electron = 1.6 X 10-19C) • What is the main formula we use? • What constants might give us a hint?) • E = Φ + ½ mv2 • hf = hfo + ½ mv2 • Hence Φ = hfo
Zinc is illuminated with UV light of wavelength 240nm. The work function is 4.3 eV. Calculate the threshold frequency of zinc and Max KE of emitted electrons • Data above has to be converted into forms that we can use. • 240 nm…………..240 X 10-9 m • The work function 4.3 eV needs to be converted into Joules……….JDeV………..4.3 eV becomes 4.3 X (1.6 X 10-19) = 6.4 X 10-19 Joules
Zinc is illuminated with UV light of wavelength 240nm. The work function is 4.3 eV. Calculate the threshold frequency of zinc and Max KE of emitted electrons • E = Φ + ½ mv2 • hf = hfo + ½ mv2 • Hence Φ = hfo • fo = Φ/h = 6.4 X 10-19J/ 6.6 X 10-34Js • fo = 0.97 X 10-15 Hz
Zinc is illuminated with UV light of wavelength 240nm. The work function is 4.3 eV. Calculate the threshold frequency of zinc and Max KE of emitted electrons • E = Φ + ½ mv2 • hc/λ = hfo + ½ mv2 • (6.6 X 10-34)(3 X 10-8) = 6.4 X 10-19 + ½ mv2 240 X 10-9 • (6.6 X 10-34)(3 X 10-8) = ½ mv2 (KE) (240 X 10-9)(6.4 X 10-19)
An electron accelerates from the cathode through a P.D. of 4kV in a CRT. How much energy does the electron gain? What is the speed of the electron at the anode? • As usual we see that they are looking for the energy gained • eV = ½mv2 • eV = (1.6 X 10-19)(4000) = 6.4 X 10-16 Joules = KE
An electron accelerates from the cathode through a P.D. of 4kV in a CRT. How much energy does the electron gain? What is the speed of the electron at the anode? • eV = ½mv2 • 6.4 X 10-16 Joules = ½mv2 • 6.4 X 10-16 Joules = ½(9.1 X 10-31)v2 • (6.4 X 10-16)(2) = v2 (9.1 X 10-31) • V = 1.18 X 108 ms-1
After leaving the anode, the electron travels at a constant speed and enters a magnetic field at right angles where it is reflected. The flux density is 5 X 10-2 T. Calculate the force acting on the electron? • F = qvB • F = (1.6 X 10-19)(1.18 X 108)(5 X 10-2) • F = 9.44 X 10-13 N
After leaving the anode, the electron travels at a constant speed and enters a magnetic field at right angles where it is reflected. The flux density is 5 X 10-2 T. Calculate the radius of the circular path followed by the electron in the field? • F = qvB F = mv2/r • qvB = mv2/r • qvBr = mv2 • qBr = mv • r = mv/qB
After leaving the anode, the electron travels at a constant speed and enters a magnetic field at right angles where it is reflected. The flux density is 5 X 10-2 T. Calculate the radius of the circular path followed by the electron in the field? • r = mv/qB • r = (9.1 X 10-31)(1.18 X 108) (1.6 X 10-19)(5 X 10-2) r = 1.34 X 10-2 m
Radium-226 undergoes α-decay and has a decay constant of 1.35 X 10-11s-1. Calculate the number of α-particles emitted per second by a 2μg sample of this isotope.(1 mol of radium-226 is 226 g; Avagadros constant = 6.02 X 1021 mol-1) • 1 mol = 226 g = 6.02 X 1021 atoms • 1g =6.02 X 1021/226 = 2.66 X 1019 atoms • 1μg =2.66 X 1019 / 106= 2.66 X 1013 atoms • 2μg = 5.32 X 1013 atoms
Radium-226 undergoes α-decay and has a decay constant of 1.35 X 10-11s-1. Calculate the number of α-particles emitted per second by a 2μg sample of this isotope.(1 mol of radium-226 is 226 g; Avagadros constant = 6.02 X 1021 mol-1) • Number of α-particles = λN • λN = (5.32 X 1013)(1.35 X 10-11) • No. of α-particles = λN = 7.182 X 102 particles
A detector records 1200 counts per minute when the activity of a radioactive sample is first measured. Six minutes later the activity has fallen to 150 counts per minute. Calculate the half-life of the sample • 1200 600 300 150 1 half-life2 half-lives3 half-lives • 3 half lives in 6 min…Each half-life in 2 min
An ancient wooden cup from an archaeological site has an activity of 2.1 Bq. The corresponding for newly cut wood is 8.4 Bq. If the half-life of Carbon-14 is 5730 years, estimate the age of the cup. • 8.4Bq 4.2Bq 2.1Bq 1 half-life2 half-lives • 2 half lives each half-life being 5730 years each…….11460 years
The power generator in a nuclear reactor is 150MW. Calculate the number of fissions occurring per second in the reactor, give that 180MeV of energy is released per fission • Firstly, get all units into SI units. • 150 MW is 150 X 106 Watts • 180MeV is 180 X 106 eV but this isn’t an SI • 180 x 106 eV…….…..…JDeV…………….. (180 x 106)(1.6 X 10-19) = 2.88 X 10-11 Joules
The power generator in a nuclear reactor is 150MW. Calculate the number of fissions occurring per second in the reactor, give that 180MeV of energy is released per fission • Watts = 150 X 106 Watts • Watts = Energy per Second • Energy released per fission = 2.88 X 10-11 J • Fissions per Sec = Energy/ Energy released per fission • Fissions per Sec = (150 X 106)/(2.88 X 10-11) = 4.32 X 10-3 fissions per sec
If a sample of radium contains 2.6 X 1021 radium-226 and is emitting 3.5 X 1010 particles per second, calculate the decay constant • Rate of decay = λN • Rate of decay/N = λ • 3.5 X 1010/ 2.6 X 1021 = λ • λ = 1.35 X 10-11 s-1
If a sample of radium contains 2.6 X 1021 radium-226 and is emitting 3.5 X 1010 particles per second, calculate the half-life if the rate of decay is 1.35 X 10-11 s-1 • T½ = 0.693/λ • T½ = 0.693/1.35 X 10-11 • T½ = 5.15 X 1010 s
A large number of solar panels are joined together in series and cover an area of 20m2. The efficiency of the solar cells is 20%. If the solar cell constant is 1400 Wm-2, what is the max power generated by the solar cells • Because the efficiency is only 20% on 20m2, it’s like having only 4m2 solar cells. • Solar constant is 1400 Wm-2. From the unit I see that this is power/ area
A large number of solar panels are joined together in series and cover an area of 20m2. The efficiency of the solar cells is 20%. If the solar cell constant is 1400 Wm-2, what is the max power generated by the solar cells • Solar constant is 1400 Wm-2. From the unit I see that this is power/ area. • 1400 = Power/ area • 1400 X area = Power • 1400 X 4 = Power = 5600W
Cobalt-60 is a radioactive isotope with a half-life of 5.26 years and emits β-particles. Write an equation to represent the decay of cobalt-60. • Cobalt-60 ---------- β + substance-61
Cobalt-60 is a radioactive isotope with a half-life of 5.26 years and emits β-particles. Calculate the decay constant of cobalt-60. • T½ = 0.693/λ • λ= 0.693/T½ • λ = 0.693/5.26 • λ = 0.13175 y-1
Cobalt-60 is a radioactive isotope with a half-life of 5.26 years and emits β-particles. Calculate the rate of decay of cobalt-60 when it has 2.5 X 1021 atoms. Rate of decay = λN Rate of decay = λ X N Rate of decay = 0.13175/ 2.5 X 1021 Rate of decay = 0.33 X 1021 Rate of decay = 3.3 X 1020 particles
A neutral pion is unstable with a decay constant of 2.5 X 1012 s-1. What is the half-life of a neutral pion? • T½ = 0.693/λ • T½ = 0.693/2.5 X 1012 • T½ = 2.772 X 10-13 s