Electron Transport Chain
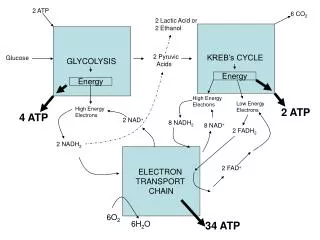
Electron Transport Chain. Review. Glycolysis & the Krebs Cycle only produce 4 ATP/glucose Most of the energy from glucose is stored in NADH or FADH 2. Location. The Electron Transport Chain (ETC) takes place along the Inner Mitochondrial Membrane. Components.

Electron Transport Chain
E N D
Presentation Transcript
Review • Glycolysis & the Krebs Cycle only produce 4 ATP/glucose • Most of the energy from glucose is stored in NADH or FADH2
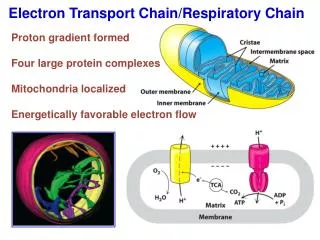
Location • The Electron Transport Chain (ETC) takes place along the Inner Mitochondrial Membrane
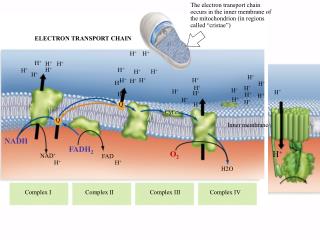
Components • Most components of the Electron Transport Chain are protein complexes • These complexes have Prosthetic Groups • Essential non-protein components
The entire Electron Transport Chain consists of 4 transmembrane structures as well as a pair of electron carriers • Most structures are made up of more than one protein
NADH Dehydrogenase(Complex I) • NADH is oxidized and in turn reduces flavin mononucleotide (FMN) • FMN participates in another redox reaction with an Iron-Sulfur protein • FMN is oxidized, Fe•S is reduced
First Electron Carrier • Fe•S part of NADH Dehydrogenase pass the electrons to Ubiquinone (Q) • Lipid electron carrier • Transports the electrons to Cytochrome b-c1 complex
Cytochromeb-c1 Complex (Complex III) • Cytochrome b (Cyt b) accepts the electrons from Q • Electrons are passes from Cyt b to another iron-sulfur protien before being passed to Cytochrome c1
Second Electron Carrier • Cytochrome c1 passes the electrons to the second electron carrier, Cytochrome c • Cytochrome c operates on the outside of the inner mitochondrial membrane (in the intermebrane space)
CytochromeOxidase Complex (Complex IV) • Consists of two protein complexes: Cytochrome a and Cytochrome a3 • Cytochrome a receives the electrons from Cytochrome c and then transfers electrons to Cytochrome a3 • Cytochrome a3 passes the electrons onto Oxygen • Combines with hydrogen atoms in the Matrix to form water
Each transfer of electrons is an exothermic redox reaction • The energy is used to transfer a single H+ ion into the Intermembrane Space • Oxygen (due to its high electronegativity) is the only element available to accept electrons at the end
FADH2 transfers its electrons to a lower energy level than NADH • Electrons join up with Ubiquinone (Q) hence NADH 3 H+ moved FADH2 2 H+ moved FADH2 FAD
Chemiosmosis • The final stage of Oxidative Phosphorylation • The complexes of the ETC create a electrochemical gradient of H+ across the inner membrane • H+ want to return to equilibrium
ATP Synthase • Protein imbedded in the inner mitochrondia membrane • Allows H+ ions back through into the matrix • Uses this Proton-Motive Force to convert ADP and ATP • Each H+ ion let back into the matrix creates 1 ATP
Energy Tally/Glucose Molecule Glycolysis 2 ATP Krebs Cycle 2 ATP ETC NADH (Glycolysis) 4-6 ATP NADH (Prep Step) 6 ATP NADH (Krebs) 18 ATP FADH24 ATP 36-38 ATP