Understanding Isotopes: Nature, Stability, and Radioactivity
Isotopes are atoms of the same element with varying numbers of neutrons. While they share the same number of protons, their mass numbers differ. Isotopes can be classified as natural or man-made, and stable or unstable. Stable isotopes do not undergo radioactive decay, whereas unstable isotopes, known as radioisotopes, do, releasing subatomic particles. Examples include Carbon-12, Carbon-14, Helium-3, and Helium-4. Understanding isotopes is crucial in fields like nuclear chemistry, medicine, and environmental science.

Understanding Isotopes: Nature, Stability, and Radioactivity
E N D
Presentation Transcript
- Composition NotebookIsotopes are atoms of the same element that have a different number of neutrons in their nuclei.
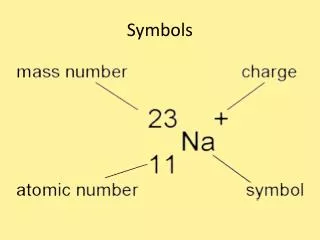
Protons make the element what it is ! • Isotopes of an element will have the same number of protons but a different number of neutrons. • The number of protons in the nucleus of an atom and is the same for all atoms of the same element.
Mass Number • is the number of protons + the number of neutrons in the nucleus of an atom and will be different for each isotope of an element.
Isotope classification • Isotopes can be classified as natural (found in nature) or man made (artificial or synthetic). • Isotopes can also be classified as stable or unstable
Stable vs. Unstable • A stable isotope does NOT undergo radioactive (or nuclear) decay. • An unstable isotope undergoes radioactive (or nuclear) decay. Unstable isotopes are also known as radioisotopes or radionucleides
Examples of Isotopes- atoms with different numbers of neutrons- naturally occurring isotopes • Hydrogen- 1 and Hydrogen -2 • Helium-3 and Helium-4 (one extra neutron) • Lithium-3 and Lithium-7 (4 extra neutrons) • Boron-10 and Boron-11 (one extra neutron) • Carbon-12, Carbon-13, Carbon-14 • Nitrogen-14 and Nitrogen-15 • Oxygen-16, Oxygen-17, and Oxygen-18 • Neon- 20, Neon-21, and Neon-22
Unstable Isotopes • Carbon-14 (two more neutrons than C-12) • Uranium-234, Uranium-235, Uranium-238
Practice Drawing Isotopes Helium-3 and Helium-4 (one extra neutron) Lithium-3 and Lithium-7 (4 extra n
Practice Drawing Isotopes Lithium-3 and Lithium-7 (4 extra neutrons) Boron-10 and Boron-11 (one extra neutron) Carbon-12, Carbon-13, Carbon-14
Radioactivity • A nucleus of an atom is unstable and releases subatomic particles. • Radioactivity is a random process, meaning that it is physically impossible to predict whether or not a given atomic nucleus will decay and emit radiation at any given moment
United Streaming ResourcesRadioactivity: Nuclear Disintegration and subatomic particlesRadium and Radioactivity 2 min clipWelcome to Discovery Education Player
Follow Up Questions • What is an isotope? • What subatomic particle makes an atom an isotope? • Do isotopes have different numbers of protons?
Follow Up Questions • What is radioactivity? • Where does radioactivity happen in the atom? • What is released when an atom decays? • What happens to the atom when it decays? Does it change?