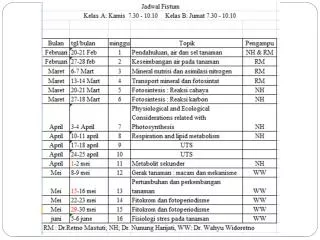
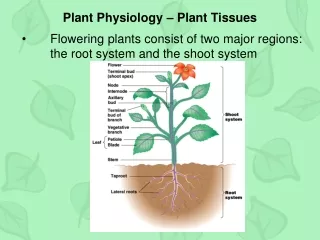
Plant Physiology
Plant Physiology. Water and Plant Cells. Water and plant cells I. Background on water in plants II. The properties of water III. Understanding the direction of water movement: Water potential. Water Plant cells are mostly water; 80 - 95% of the mass of growing cells,

Plant Physiology
E N D
Presentation Transcript
Plant Physiology Water and Plant Cells
Water and plant cells I. Background on water in plants II. The properties of water III. Understanding the direction of water movement: Water potential
Water • Plant cells are mostly water; 80 - 95% of the mass of growing cells, Wood (Sapwood 35-75%) • Seeds 5-15% • Living cells must maintain a positive water pressure, or “turgor” to grow and function properly. Cell walls build Internal Hydrostatic Pressure Turgor Pressure Cell enlargement Gas Exchange in Leaves • Transport in Phloem • Transport across membrane Rigidity and Mechanical stability to nonlignified plant tissues
Water • Plants lose large quantities of water in transpiration, the evaporation from the interior of leaves through the stomata is referred to as Transpiration. • CO2 H2O • 100% water exchange in one hour • Water loss through leaf surface • Heat dissipation by leaves through transpiration • Half of the heat by sunlight is dissipated by transpiration • Latent Heat of Vaporization
Plant - Water Relations Corn yield as a function of water availability
Plant - Water Relations Productivity of various ecosystems as a function of annual precipitation
Water passes easily throughbiological membranes, particularly through Aquaporins - low resistance pores. Formed by Aquaporins Aquaporins: are proteins embedded in cell membrane that regulate flow of water. Prof Peter Agre and discovery of Aquaporins?
II. The properties of water Polar molecule that forms hydrogen bonds. 1) good solvent 2) cohesive properties - attraction to like molecules 3) adhesive properties - attraction to unlike molecules The polarity of water molecules results in hydrogen bonding Covalent bond
Properties of water, continued • Cohesion is the attraction of like molecules (H2O here) that gives water its tensile strength. • Adhesion is the attraction of unlike molecules. Water adheres to cell walls, soil particles, glass tubes, etc. • Adhesion explains capillarity & surface tension. • Surface Tension is caused by cohesive forces within liquid molecules. • All of above forces give rise to a phenomenon called Capillarity (the movement of water along a capillary tube)
III. What factors determine the direction of water movement (through the soil, between cells, from roots to leaves, from leaves into air)? • Gravity 2. Pressure 3. Concentration
100 90 water flows upward in trees. How does this work? 80 70 Height, meters 60 50 40 Gravity causes water to move downward unless it is opposed by an equal and opposite force. 30 20 10 0
Pressure Water moves from regions of higher to lower pressure garden hose straw through xylem of plants
Water pressures in plant cells can be positive (turgor), or negative, (tension). Living cells ≥ 0 MPa to ≈ +3 MPa) Dead xylem cells ≤ 0 MPa, to as low as -12 MPa.
3) Concentration Water moves by diffusion from regions of higher to lower water concentration. Solutes added to pure water dilute the water concentration.
Osmosis is the diffusion of water across a selectively permeable membrane from a region of higher to lower water concentration. How does reverse osmosis purify water? A process by which a solvent passes through a porous membrane in the direction opposite to that for natural osmosis.
The concept of water potential, Y, brings together the influences of gravity, pressure, and concentration (solutes) in describing the energy state of water and the direction of water movement. The water potential equation: YW = YS + YP + Yg YW = total water potential YS = solute potential YP = pressure potential Yg = gravitational potential All units will be pressure, pascals, Pa. MPa is megapascal, 106 Pa
Change in water status causes physiological changes Measure of the rate of passage of CO2 entering or water vapors exiting through stomata
Water and Plant Cells, Chapter 3, Plant Physiology by Taiz and Zeiger Further readings