Genome Control
Genome Control. Bacterial chromosome. 1 circular chromosome Virus < bacteria < eukaryotes (# of bases) Nucleiod region Replication proceeds in both directions Rapid reproduction Single cell to colony (10^7 to 10^8 bacteria) within 12 hours Plasmids Variation in bacteria?.


Genome Control
E N D
Presentation Transcript
Bacterial chromosome • 1 circular chromosome • Virus < bacteria < eukaryotes (# of bases) • Nucleiod region • Replication proceeds in both directions • Rapid reproduction • Single cell to colony (10^7 to 10^8 bacteria) within 12 hours • Plasmids • Variation in bacteria?
Transduction: Phage carries genes from one bacterium to another Defective phage
Conjugation: And you thought bacteria couldn’t have sex? F factor Exist as part of chromosome (episome) or as a plasmid (contains mostly genes for making sex pili F+ F+ and F+ F- F- and F- During conjugation F+ F+ F- F+ Hfr – F factor on chromosome Genes transferred depends on initiation and termination
R plasmids • Function of natural antibiotics? • Advantage of antibiotic resistant genes • Consequence of prescribing antibiotics?
Insertion sequences Only in bacteria Transposase recognizes the inverted repeats May disrupt coding sequence or regulatory genes Transposons More complex (extra genes) Can add antibiotic resistance genes to plasmi already carrying genes. How would this spread? Found in eukaryotic genomes as well Transposable elementsRecombination within a genome (between chromosome and plasmid or between plasmids
Transformation • Uptake of exogenous DNA, resulting in newly acquired traits • Must be in competency (state able to take in DNA facilitated by membrane bound proteins • Competency is induced in E. Coli with CaCl2, MgCl2, or RbCl, and sudden changes of heat and cold (makes cell membrane permeable) • How do the Griffith and Avery experiments relate to transformation?
Transformation efficiency • Amount of cells transformed/μg DNA • Through selection, colonies are counted • Each colony grew from a transformed cell
pUC8 Plasmid • Present in E. Coli • Replicates independent of bacterial chromosome • Genetically engineered • Lac Z gene (β galactosidase) • MCR (can facilitate insertion of DNA (not required for replication)
pUC8 plasmid • With Inducers (IPTG and X-Gal) colonies of bacteria appear blue • Lac Z β galactosidase X-Gal cleaved blue product • With inserted gene, lac Z is interrupted and colonies appear white • No Lac Z No β galactosidase X-Gal not cleaved white product
pUC8 plasmid • Engineered E. Coli cells only synthesizes carboyxl terminal of β galactosidase protein • pUC8 plasmid contains gene for amino terminal • If pUC8 transforms cells, gene is fully functional
Further selection • E. Coli NOT resistant to antibiotic ampicillin • pUC8 contains ampicillin resistant gene • The enzyme B-lactamase exits cell and inactivates ampicillin • Satellite colonies can appear around blue colonies (which color would they be , why?)
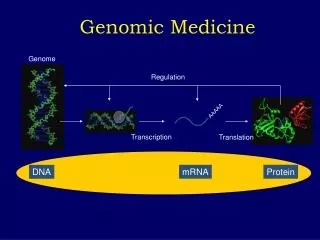
Overview of Genome Control • Gene expression must respond to external cues • Differentiation allows cells to become specialized • Expression usually regulated at transcription level by DNA-binding proteins
Prokaryotic Coiled and looped with protein (chromatin) Eukaryotic More complex Highly condensed and coiled during metaphase around histones (4 + H1) Histones leave only during DNA replication (what about transcription?) Heterochromatin vs. euchromatin Genome packing (1st level)
- Charged phosphates + charged a.a. Non histones
Chromatin modification • Chromatin function • Compact DNA • Control transcription • Euchromatin vs. heterochromatin • Gene’s location relative to nucleosome and scaffold • DNA methylation – inactivates genes • Histone acetylation – increase gene expression by changing conformation of histones
Distal Proximal Recognizes TATA box Control elements (noncoding DNA) -Transcription will occur, but inefficiently without activators -Repressors can bind to silencers to repress transcription (methylation) Recognizes proteins, including RNA Polymerase
Transcription factors • DNA-binding domain (helix-turn-helix, zinc finger, leucine zipper) • Protein binding domain
Coordinately controlled genes • Similar control elements before each gene allows simultaneous gene expression (analogous to operons) • steroid hormones and growth factors can act as signals to control expression
Posttranscriptional control • Alternative splicing • mRNA degradation (hemoglobin example) • Poly(A) tail shortened 5’ cap removed mRNA degraded (enzymatic action) • mRNA stability controlled by part of sequence near 3’ end • Translational control - Preventing mRNA attachment to ribosome (could help store mRNA for later use)
Post translational control • Protein modification • Cleavage • Chemical modification • Transportation • Posttranslational degradation
Relevant tutorials • Campbell site – 19D, 19E, 19F • http://highered.mcgraw-hill.com/sites/0072437316/student_view0/chapter18/animations.html# • Transcription Complex and Enhancers (586.0K) • Control of Gene Expression in Eukaryotes (959.0K)
Cancer caused by genetic changes All result in oncogenes “cancer genes” Agents of cancer – Spontaneous mutation, chemical carcinogens, radiation, viruses
Signaling pathway that regulates cell growth oncogene Mutated tumor suppressor
Multiple mutations leading to cancer Monday – drip Oxaliplatin Tuesday - 1/2 hour drip 5-fluorouracil Take home and use leucovorin for 22 hours
Prokaryotic DNA codes for protein, tRNA, or rRNA Noncoding regions: Regulatory sequences Coding sequence along a gene is continuous Eukaryotic Only 3% coding region Noncoding regions: Regulatory regions, repeated sequences, introns Multigene families Organization at the DNA level
Repetitive DNA - Different nucleotide composition, making the density distinct (appears as a satellite band) - Fragile X, Huntington’s - Near centromeres, telomeres Alu elements -5% of genome -300 pairs long -Transcribed into RNA - Unknown function
Gene Families • Almost like extensive repetitive DNA • Some consist of identical genes, tandemly clustered • Most code for RNA -lack regulatory sequences -Higher affinity for oxygen -Transcription separated temporally
Gene amplification • Higher rate of transcription during early development
Genome rearrangement • Transposons - Stretches of DNA that can move within a genome • Can interrupt gene expression • Can carry a gene that becomes expressed when inserted next to a promoter • Retrotransposon – needs RNA intermediate (Alu elements) • 50% of maize, 10% of humans
Relevant tutorials • Campbell site – 19A, 19B
Getting cloned eukaryotic gene into a prokaryotic setting Introns Eukaryotic versus prokaryotic genome control Engineer cloning vector to include prokaryotic promoters Extract mature mRNA and make cDNA Use yeast cells, artificial chromosomes Problems cloning DNA
PCR of VNTRs • Extract DNA from sample • Lyse cell membranes to release DNA • Add nucleotides, primers, Taq polymerase to DNA sample • Thermocycler amplifies DNA • Gel electrophoresis of amplified DNA
Sample results ladder • Will this be enough evidence to convict someone matching the DNA sample? More repeats homozygous heterozygous
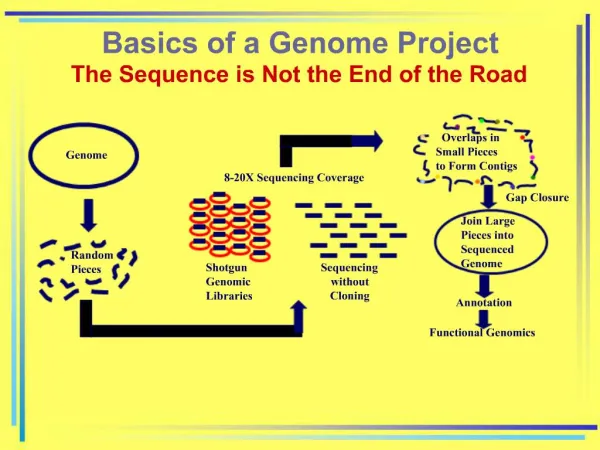
Mapping a genome • Linkage mapping • Based on recombinant frequencies • Physical mapping (chromosome walking) • DNA sequencing
Relevant tutorials • Campbell site – 19G, 20F • http://highered.mcgraw-hill.com/sites/0072437316/student_view0/chapter20/animations.html • How Tumor Suppressor Genes Block Cell Division (694.0K)
Group presentations • 1 – Genomic and cDNA libraries (367-368) • 2 – Gel Electrophoresis and RFLPs (372-374) • 3 – Southern Blotting (372, 373, 375) • 4 – Sanger Method (378) • 5 – DNA microarray assays and in vitro mutagenesis (379)
Relevant tutorials • Campbell site: 20B, 20D, 20F • http://highered.mcgraw-hill.com/sites/0072437316/student_view0/chapter16/animations.html