THE ATOM
THE ATOM. Around 400 B.C. a Greek philosopher, Democritus , proposed that all forms of matter were divisible into extremely small fundamental particles called atomos . (Greek for indivisible)


THE ATOM
E N D
Presentation Transcript
THE ATOM • Around 400 B.C. a Greek philosopher, Democritus, proposed that all forms of matter were divisible into extremely small fundamental particles called atomos. (Greek for indivisible) • Due to world events, this concept was forgotten, replaced by the belief that matter was composed of four “elements” ; earth,wind, fire, & water. • From 1803 to 1810, John Dalton proposed the concept of the atom once again. Dalton backed his claim with experimentation.
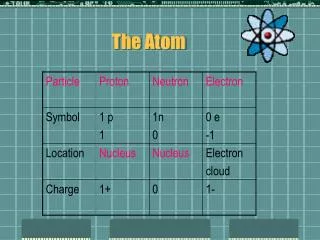
Subatomic Particles • ELECTRON: Properties discovered in 1897 by JJ Thompson • PROTON: Isolated and identified in 1919 by Ernest Rutherford • NEUTRON: Discovered by James Chadwick in 1932
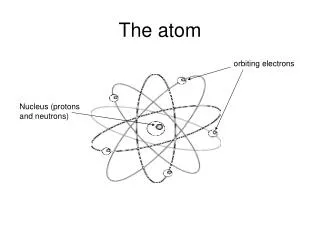
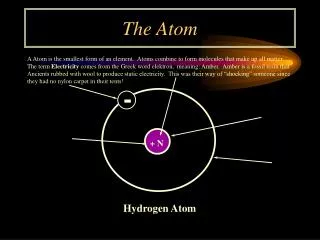
Nuclear Model of the Atom • Every atom contains an extremely small, extremely dense nucleus. • All of the positive charge and nearly all of the mass of an atom are concentrated in the nucleus. • The nucleus is surrounded by a much larger volume of nearly empty space that makes up the rest of the atom. • The space outside the nucleus is very thinly populated by electrons, the total charge of which exactly balances the positive charge of the nucleus.