Positively charged ions are called cations (named first)
400 likes | 615 Vues
Positively charged ions are called cations (named first) Negatively charged ions are called anions Cations can be metals or polyatomic ions

Positively charged ions are called cations (named first)
E N D
Presentation Transcript
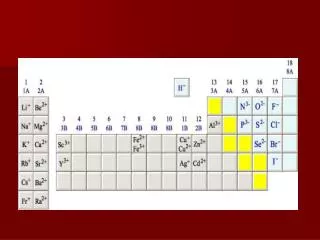
Positively charged ions are called cations (named first) • Negatively charged ions are called anions • Cations can be metals or polyatomic ions • For metals that have only one possible charge (valency) the name of the metal is used.Examples are Group I metals (charge 1+), Group II metals (charge 2+), Aluminium (charge 3+), Zinc (charge 2+), Silver (charge 1+) • For metals that can have more than one charge (valency) the name of the metal is succeeded by the valency in capital Roman numerals in bracketsORby using the suffix -ous for the lowest valency and -ic for the highest valency and sometimes with the Latin name for the metal
Anions can be a negatively charged element or a polyatomic ion • Negatively charged elements have the suffix -ideExamples are oxide (O2-), sulfide (S2-), fluoride (F-), chloride (Cl-), bromide (Br-), iodide (I-), nitride (N3-), hydride (H-) • Polyatomic ions which include oxygen in the anion have the suffixes -ate or -ite. "ate" means there is more oxygen in the anion than one ending in "ite"Examples: sulfate (SO42-) has more oxygen than sulfite (SO32-), nitrate (NO3-) has more oxygen in the anion than nitrite (NO2-)Other examples are carbonate (CO32-), phosphate (PO43-) and permanganate (MnO4-)Exception: OH- is named hydroxide
MgO CATION: Mg2+ is named magnesium as magnesium belongs to Group (II) and can only have one charge (valency)ANION: O2- is named oxideName of compound is magnesium oxide
FeS CATION: Fe2+ is named iron (II) or ferrous as iron can have a charge of either 2+ or 3+ANION: S2- is named as sulfideName of compound is iron (II) sulfide or ferrous sulfide
LiH CATION: Li+ is named as lithium since lithium is a Group I metal and can have only one charge (valency)ANION: H- is named as hydrideName of compund is lithium hydride
CaCO3 CATION: Ca2+ is named as calcium (since it is a Group II metal) and can only have 1 chargeANION: CO32- is named as carbonateName of compound is calcium carbonate
FeSO4 CATION: Fe2+ named as iron (II) or ferrousANION: SO42- named as sulfate
FeSO3 CATION: Fe2+ named as iron (II) or ferrousANION: SO32- named as sulfiteName of compound is iron (II) sulfite or ferrous sulfite
Guided Practice • Ca I2 =
Guided Practice • Ca+2 I2-1 =
Guided Practice • Ca+2 I2-1 = CaI2
Guided Practice • Ca+2 I2-1 = CaI2 Calcium Iodide
Guided Practice Na2+1 CO3-2 =
Guided Practice Na2+1 CO3-2 = Na2(CO3)
Guided Practice Na2+1 CO3-2 = Na2(CO3) = Sodium Carbonate
Guided Practice • Cu F2 =
Guided Practice • Cu+2 F2-1 =
Guided Practice • Cu+2 F2-1 = CuF2 • Copper (II) Fluoride
Guided Practice • Fe SO4=
Guided Practice • Fe +2 SO4-2=
Guided Practice Fe +2 SO4-2 = Fe (SO4)
Guided Practice Fe +2 SO4-2 = Fe (SO4) = Iron (II) Sulfate
Guided Practice Iron (III) Oxide =
Guided Practice Iron (III) Oxide = Fe+3 O-2
Guided Practice Iron (III) Oxide = Fe+3 O-2 = Fe2 (Total of +6 charge)
Guided Practice Iron (III) Oxide = Fe+3 O-2 = Fe2O3
Guided Practice Copper (II) Chloride =
Guided Practice Copper (II) Chloride = Cu+2 Cl-1
Guided Practice Copper (II) Chloride = Cu+2 Cl-1 = CuCl2
Guided Practice • Ga (ClO3)3 =
Guided Practice • Ga (ClO3)3-1 =
Guided Practice • Ga+3 (ClO3)3-1 = Ga (ClO3)3
Guided Practice • Ga+3 (ClO3)3-1 = Ga (ClO3)3 = Gallium Chlorate
Guided Practice (NH4)3 (PO4)
Guided Practice (NH4)3 +1 (PO4) -3
Guided Practice (NH4)3 +1 (PO4) -3 = (NH4)3 (PO4)
Guided Practice (NH4)3 +1 (PO4) -3 = (NH4)3 (PO4) = Ammonium Phosphate
Guided Practice Mg (NO3)2 = Magnesium Nitrate = Mg (NO3)2