Spectroscopy
Nuclear Magnetic Resonance Spectroscopy. NMR Spectroscopy is the study of the interaction of electromagnetic radiation and nuclei in a magnetic fieldThe most powerful method of molecular spectroscopy, NMR allows the determination of connectivity, stereochemistry, conformations, and even dynamics o

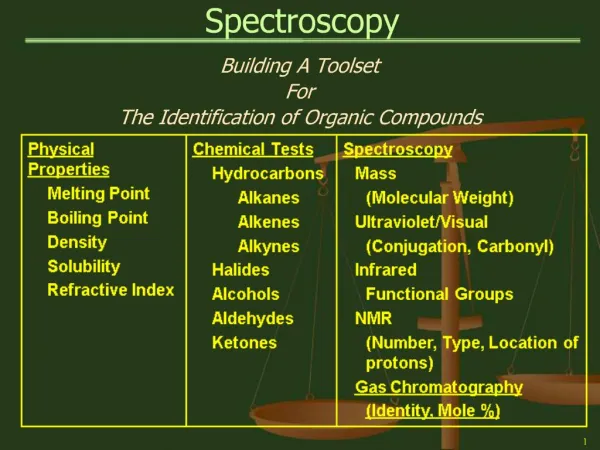
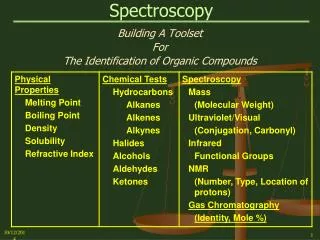
Spectroscopy
E N D
Presentation Transcript
1. Spectroscopy PART II: Nuclear Magnetic Resonance Spectroscopy - Theory and 1H NMR Spectra
Organic Chemistry 30B
2. Nuclear Magnetic Resonance Spectroscopy NMR Spectroscopy is the study of the interaction of electromagnetic radiation and nuclei in a magnetic field
The most powerful method of molecular spectroscopy, NMR allows the determination of connectivity, stereochemistry, conformations, and even dynamics of molecular structures.
3. Energy, Wavelength, Frequency
4. NMR Spectroscopy Absorption of radio waves in the wavelength range of 10 m (frequency of 330 MHz) causes nuclear spin flips
The resonance frequencies are a function of the local magnetic field which is exquisitely dependant on the molecular structure
5. Spin Nuclei have Spin Quantum Numbers
6. Spin Energies In a magnetic field different spin states have different energies
7. Spin Energies The difference in energy between the spin states is dependent on the magnitude of the applied magnetic field Bo
8. Changing Spin States
9. Resonance Frequency When a nucleus is irradiated with electromagnetic radiation at the exact frequency of precession it is in �resonance�. Energy is absorbed and the nuclear spin flips to the higher energy state. The absorption of energy is detected by the NMR spectrometer and plotted as a signal.
10. NMR Spectrometer
11. Spin Energetics The difference in energy between the spin states is dependent on the magnitude of the applied magnetic field Bo
DE = h?Bo/2?
h = Planck�s constant
? = Magnetogyric ratio
Bo = Applied magnetic field
12. Spin Energetics The difference in energy between the spin states is dependent on the magnitude of the applied magnetic field Bo
DE = h?Bo/2?
But�..
Molecules have electrons and the movement of electron density creates induced magnetic fields which can add to or subtract from the applied field.
Therefore, nuclei in different environments will have different resonance frequencies.
13. Chemical Shift The methyl protons in propane have a resonance frequency of 400,000,360 Hz at 9.4 Tesla, while the methylene protons have a resonance frequency of 400,000,520 Hz.
The exact frequencies are less important than the changes.
14. Chemical Shift Therefore, the Chemical Shift is the difference is resonance frequencies between the nucleus of interest and a reference compound, tetramethylsilane (Me4Si , TMS).
The standard unit is Parts Per Million
At 400 MHz, 1 ppm = 400 Hz
Chemical shift of TMS defined as 0 ppm
15. Chemical Shift The standard unit is Parts Per Million
At 400 MHz, 1 ppm = 400 Hz
Therefore:
The TMS signal at 400,000,000 Hz is defined as a Chemical Shift of 0 ppm
The methyl signal in propane at 400,000,360 Hz is then 0.9 ppm
The methylene signal in propane at 400,000,520 Hz is then 1.3 ppm
16. 1H NMR Spectrum
17. Chemical Shift Greater electron density creates a greater induced magnetic field to oppose the applied magnetic field. Thus the nuclei are shielded.
Reduced electron density creates a lessor induced magnetic field to oppose the applied magnetic field. Thus the nuclei are deshielded.
18. Chemical Shift Proton NMR signals near 1-2 ppm are for nuclei that are shielded.
Proton NMR signals greater than 2 ppm are for nuclei that are deshielded.
19. 1H NMR Spectrum
20. NMR Spectra
21. 1H NMR Spectra
22. 13C NMR Spectra
23. Equivalency of Atoms or Groups
24. Homotopic Homotopic nuclei or groups are interchangeable by an axis of rotation. Homotopic protons are equivalent so will present one signal AND will not couple to each other.
25. Homotopic Substitution Test: Two atoms or groups are homotopic if substitution of an atom or group by something else generates a structure that is identical to that generated by replacing the other atom or group.
26. Enantiotopic Enantiotopic nuclei or groups are interchangeable by a rotation and a reflection. Enantiotopic protons are equivalent by most NMR techniques so will presnt one NMR signal AND do not couple to each other.
27. Enantiotopic Substitution Test: Two atoms or groups are enantiotopic if substitution of an atom or group by something else generates the enantiomer of the compound generated by replacing the other atom or group.
28. Diastereotopic Diastereotopic nuclei are not related by any symmetry operations. Diastereotopic protons are nonequivalent so will present different NMR signals AND may couple to each other.
29. Diastereotopic Substitution Test: Two atoms or groups are diastereotopic if substitution of an atom or group by something else generates a diastereomer of the compound generated by replacing the other atom or group.
30. Topicity and Number of Signals The protons on a methyl group are Homotopic, so only one signal is seen, not three.
The two methyl groups on p-xylene are Homotopic, so only one signal is seen for both.
The four hydrogens on the benzene ring of p-xylene are also Homotopic, so only one signal is seen, not four.
31. Integration Integration only indicates the ratios of the number of protons
Thus p-xylene has two signals in a 2 : 3 ratio
32. Chemical Shift Molecules have electrons and the movement of electron density creates induced magnetic fields which can add to or subtract from the applied field.
Therefore, nuclei in different environments will have different resonance frequencies or Chemical Shifts.
The Chemical Shifts in H NMR provide an indication of the type of carbon attached (sp3, sp2, sp) and the substituents (ie heteroatoms) nearby.
33. Chemical Shift
34. Chemical Shift
35. Example NMR Spectrum
36. Chemical Shift
37. Chemical Shift
38. Chemical Shift
39. Chemical Shift
40. Chemical Shift
41. Chemical Shift
42. Example NMR Spectrum
43. Example NMR Spectrum
44. Example NMR Spectrum
45. Example NMR Spectrum
46. Experimental Detail - Solvent
47. Coupling
48. Coupling
49. Coupling
50. Coupling
51. Coupling
52. Coupling
53. Coupling WHAT PROTONS COUPLE?
Homotopic protons do not show coupling to each other
Enantiotopic protons do not show coupling to each other
Diastereotopic protons DO show coupling to each other
54. Example NMR Spectrum
55. Proton Exchange
56. Example NMR Spectrum
57. Example NMR Spectrum
58. Example NMR Spectrum
59. Unequal Coupling The magnitude of coupling is not always equal
60. Magnitude of Coupling Coupling is a through-bond process
Therefore, the magnitude of coupling decreases with distance
Therefore, not all couplings are equal
1J Coupling - One bond: 13C-H
Alkanes (sp3 carbon) - 125 Hz
Alkenes (sp2 carbon) - 150 Hz
Alkynes (sp carbon) - 250 Hz
61. Magnitude of Coupling Coupling is a through-bond process
2J Coupling - Two bonds Geminal Coupling of Diasterotopic protons
Up to 25 Hz
62. Magnitude of Coupling Coupling is a through-bond process
3J Coupling - Three bonds Vicinal Coupling
0 to 12 Hz depending on dihedral angle
63. Magnitude of Coupling Coupling is a through-bond process
4J or 5J Coupling - Long Range Coupling
0 to 6 Hz depending on several factors
64. Magnitude of Coupling
65. Example NMR Spectrum
66. Complex Coupling
67. NMR Spectral Problems
68. Interpreting 1H NMR Spectra Number of Signals - # of types of protons
Chemical Shifts - Proton environment
Integration - Ratios of numbers of protons
Signal Shape - Dynamics of proton environment
Signal Splitting - Number and geometry of nearby protons
The goal will be determination of the complete molecular structure
69. End