Understanding Atomic Structure: The Basics of Atoms and Subatomic Particles
This guide explores the fundamental concepts of atomic structure, focusing on the atom as the smallest particle of matter, primarily composed of empty space and existing in a neutral state. Learn about the nucleus, which contains protons and neutrons, and the electron shells where negatively charged electrons orbit. Explore the characteristics of the three primary subatomic particles: protons, neutrons, and electrons, including their charges and masses. Understand terms like atomic number, mass number, isotopes, and how ions are formed by adding or removing electrons to achieve stability.

Understanding Atomic Structure: The Basics of Atoms and Subatomic Particles
E N D
Presentation Transcript
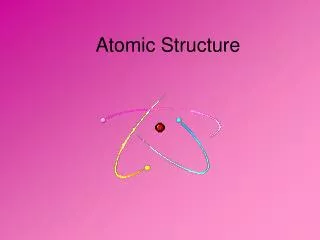
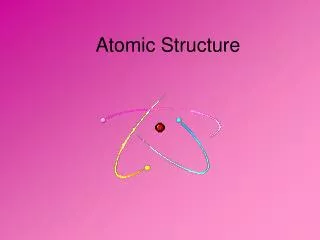
The Atom • Smallest whole particle of matter • Mostly empty space • Always neutral
Atom is Neutral • # of protons is the same as the number of electrons • Protons = + • Electrons = -
Areas of an Atom • The Nucleus • Center • Protons & Neutrons • Positively charged
Areas con’t. • Orbits or Shells • Moving electrons • Cloudlike • Negatively charged
The Subatomic Particles • Proton • Charge = +1 • Mass = 1 amu • Abbreviation = p+ • # protons = atomic number
The Subatomic Particles • Neutron • No charge • Mass = 1 amu • Abbreviation = n
The Subatomic Particles • Electron • Charge = -1 • Mass = .0005amu • Abbreviation = e-
Terms • Atomic number = # protons • Ex: carbon = 6, Boron = 5 • Mass number = # protons + # neutrons
Mass # Atomic # Isotopes • Atoms of the same element with different mass numbers. • Nuclear symbol: • Hyphen notation: carbon-12 Courtesy Christy Johannesson www.nisd.net/communicationsarts/pages/chem
Calculating Particles 11 Na 22.989 Atomic # = 11 Atomic Mass = 22.989
Calculating Particles 11 Na 22.989 11 protons (atomic #) 11 electrons (same as p+) 12 neutrons (mass #)
Making Ions • Add or remove e- • Remove = cation (+) • Add = anion (-)
Stable Atoms • Have 8 e- in the outermost orbit • The noble gases