Single and Double Replacement Reactions
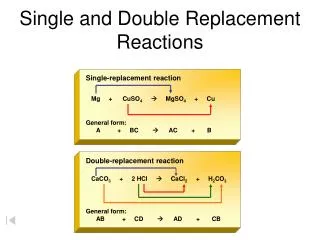
Single and Double Replacement Reactions. Single-replacement reaction. Mg + CuSO 4 MgSO 4 + Cu. General form: A + BC AC + B. Double-replacement reaction. CaCO 3 + 2 HCl CaCl 2 + H 2 CO 3.

Single and Double Replacement Reactions
E N D
Presentation Transcript
Single and Double Replacement Reactions Single-replacement reaction Mg + CuSO4 MgSO4 + Cu General form: A + BC AC + B Double-replacement reaction CaCO3 + 2 HCl CaCl2 + H2CO3 General form: AB + CD AD + CB
Single and Double Replacement Reactions Single-replacement reaction Mg + CuSO4 MgSO4 + Cu General form: A + BC AC + B Double-replacement reaction CaCO3 + 2 HCl CaCl2 + H2CO3 General form: AB + CD AD + CB