The Atom
Explore the historical development of atomic theory from Aristotle's four elements—earth, air, water, and fire—to Democritus's pioneering idea of atoms as indivisible particles. Discover John Dalton's groundbreaking atomic theory, emphasizing that different elements consist of unique atoms that combine to form compounds. Learn about key experiments, such as JJ Thompson's Plum Pudding Model and Ernest Rutherford's Gold Foil Experiment, which further refined our understanding of atomic structure, including atomic number, mass number, and isotopes.


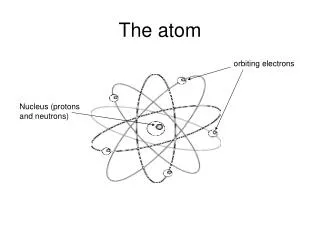
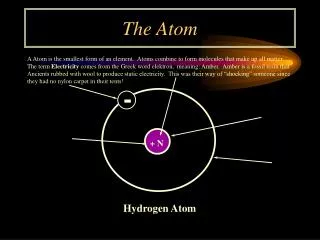
The Atom
E N D
Presentation Transcript
Aristotle – the four basic elements -Earth -Air -Water -Fire
Democritus • Follower of Leucippus • An Atomist • Coined the word atomos meaning uncuttable or the smallest particle of matter
2000 years later John Dalton – School Teacher Five main points of Dalton's atomic theory -The atoms of a given element are different from those of any other element; the atoms of different elements can be distinguished from one another by their respective relative atomic weights. -All atoms of a given element are identical. -Atoms of one element can combine with atoms of other elements to form chemical compounds; a given compound always has the same relative numbers of types of atoms. -Atoms cannot be created, divided into smaller particles, nor destroyed in the chemical process; a chemical reaction simply changes the way atoms are grouped together. -Elements are made of tiny particles called atoms.
Plum Pudding Model of the Atom JJ Thompson
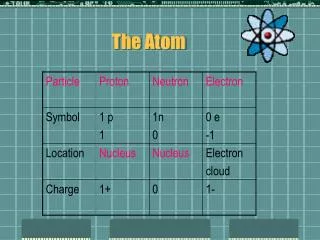
Atomic Number: The number of protons in the nucleus of an atom Mass Number: The total number of protons and neutrons on a nucleus Atomic Number Symbol Atomic Mass
Isotope: Atoms of the same element that have different masses