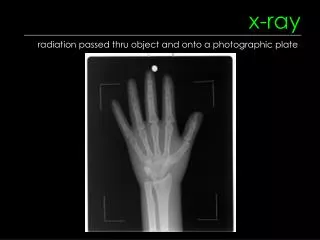
X-ray Production
X-ray Production. kVp KINETIC ENERGY OF ELECTRONS. CATHODE --------. MADE OF TUNGSTEN + 1%-3% THORIUM. TUNGSTEN. Z # 74 MELTING POINT- 3,410 DEG. CELSIUS . THORIUM. Z # 90. THERMIONIC EMISSION. CATHODE HEATED UP TO AT LEAST 2,200 DEG. CELSIUS. ANODE +++++. TUNGSTEN

X-ray Production
E N D
Presentation Transcript
CATHODE -------- MADE OF TUNGSTEN + 1%-3% THORIUM
TUNGSTEN Z # 74 MELTING POINT- 3,410 DEG. CELSIUS
THORIUM Z # 90
THERMIONIC EMISSION CATHODE HEATED UP TO AT LEAST 2,200 DEG. CELSIUS
ANODE +++++ TUNGSTEN TARGET
TUNGTEN AS TARGET HIGH Z# - 74------EFFICIENCY OF X-RAY PRODUCTION HIGH MELTING POINT –3,410 ° C– TARGET HEATED TO 2,000° C
X-RAY PRODUCTION • BREMSSTRAHLUNG RADIATION • CHARACTERISTIC RADIATION
BREMSSTRAHLUNG RADIATION If an incoming free electron gets close to the nucleus of a target atom, the strong electric field of the nucleus will attract the electron, thus changing direction and speed of the electron. The Electron looses energy which will be emitted as an X-ray photon. The energy of this photon will depend on the degree of interaction between nucleus and electron, i.e. the passing distance. Several subsequent interactions between one and the same electron and different nuclei are possible. X-rays originating from this process are called bremsstrahlung. Bemsstrahlung is a German word directly describing the process: "Strahlung" means "radiation", and "Bremse" means "brake
DIFFERENT DEGREES OF DECCELERATION X-RAYS HEAT
BREMS RADIATION IS: POLYENERGETIC ! 90% OF X-RAYS ARE PRODUCED THROUGH BREMS INTERACTIONS WHEN 80-100 KVP APPLIED
Characteristic X-rays The high energy electron can also cause an electron close to the nucleus in a metal atom to be knocked out from its place. This vacancy is filled by an electron further out from the nucleus. The well defined difference in binding energy, characteristic of the material, is emitted as a monoenergetic photon. When detected this X-ray photon gives rise to a characteristic X-ray line in the energy spectrum.
Characteristic Radiation KE OF PROJECTILE ELECTRON > BINDING ENERGYORBITAL ELECTRON
TUNGSTEN-74 BINDING ENERGIES OF DIFFERENT SHELL ELECTRONS K-70 KEV L-12 KEV M-2.8 KEV
CHARACTERISTIC X-RAYS L K 70-12 = 58 keV 70-3 = 67 keV M K M 12-3 = 9 keV L
EXCITATIONS AND TRANSFERS OF ELECTRONS BETWEEN OUTER SHELLS RESULTS IN: HEAT PRODUCTION !
EACH CHARACTERISTIC RADIATION ( ex. K TO L TRANSFER) IS: MONOENERGETIC ! THERE ARE MANY CHARACTERISTIC RADIATION PRODUCED IN ONE ATOM THEREFORE CHARACTERISTIC RADIATION IS ALSOPOLYENERGETIC !
X-RAYS PRODUCTION IS AN INEFFICIENT PROCESS 80 KVP 99.4% HEAT 0.6% X-RAYS
% EFFICIENCY OF X-RAY PRODUCTION % EFFICIENCY= k x Z x kVp k = 1 x 10-4 or 0.0001
X-RAY EMISSION • ISOTROPICAL- IN EVERY DIRECTION