Nuclear Energy
Nuclear Energy. How it works and how the world sees it today By: Harsimar Virdi, Bharti Gupta, Darren Tan, Mani Akunuri, Sara Zhu, Pardeep Brar and Siddharth Ramesh. Introduction.

Nuclear Energy
E N D
Presentation Transcript
Nuclear Energy How it works and how the world sees it today By: Harsimar Virdi, Bharti Gupta, Darren Tan, Mani Akunuri, Sara Zhu, Pardeep Brar and Siddharth Ramesh
Introduction • As a group we decided to choose this topic because not only does it incorporate the 3 sciences but it is a highly controversial topic • There are many aspects to Nuclear Energy both societal, ethically, morally, environmentally and economically • We complied this PowerPoint to show you, the audience, what we have discovered • A more condensed version of this was also created and given to the public • We then asked the public what they thought about Nuclear Energy (the new wave of the future? Or not?) • Each person was given the condensed version (brochure) illustrating what Nuclear Energy is, the pros, and the cons • Through discussions among our own group, and with the consideration of public opinion here is what we have discovered:
Table of Contents 4. Nuclear Energy 5. Nuclear Fission vs. Nuclear Fusion 6. Mass energy Formula 7-9. Nuclear Fission 10-12. Nuclear fusion 13. How it works video 14. the Uranium Atom 15. Uranium in Nuclear Energy 16-17. How to get Uranium 18. Radiation and Nuclear Reactions 19-20. Types of Radiation 21. Nuclear Power Plants 22. Nuclear Reactors 23-28. Types of Nuclear Reactors 29-30. Safety: Maintaining a Nuclear Reactor 31. Safety of the Workers 32-33. Workplace: Accidents that have occurred 34. Nuclear Waste 35. Mutations 36. Chromosomes 37. Table 1: Radiation Dose 38-39. Timeline: progression of Nuclear Energy 40. Uses of Nuclear Energy 41. Benefits of Radioisotopes 42. Increasing Demand for Nuclear Energy 43. Nuclear Energy Today 44. Table 2: World Energy Usage During 2007 45-46. Social/Economical Benefits to Nuclear Energy 47. Risks of Nuclear Energy 48. Weapon of Mass Destruction? Video 49. The Survey 50-52. Survey Results 53-55. Findings 56. Strengths 57. Weaknesses 58. Possible Improvements 59. Conclusion 60-61. Bibliography
Nuclear Energy Nuclear Energy is a growing form of energy production and is retrieved through the use of FUELS such as Uranium-235. The fuel is not burned, but rather energy is achieved through FISSION REACTION Like any type in energy it has its advantages and disadvantages - its clean, reliable and affordable - however it does deal with radiation hazards and is nonrenewable source: Greenpeace 2003 http://incentraleurope.radio.cz/pictures/eu/cd/energetika/energie_jaderna.jpg
Nuclear Fission vs. Nuclear Fusion Nuclear energy is released through either nuclear fission or fusion • Nuclear Fission: • This occurs when energy is released after a nucleus splits apart • It usually splits into 2 pieces, but sometimes 3 as well • Nuclear Fusion: • Energy is released when two nuclei merge into 1 NASA. US-HUBBLE-PLANET-ARTISTS CONCEPT. Agence France Presse. 27 Nov 2001.
Mass Energy Formula • To describe nuclear energy there is the mass-energy equivalence formula: ΔE = Δmc² • In this formula E= energy that is released, m= mass defect and c= speed of light • Derived by Albert Einstein, who proved that since the speed of light in a vacuum is a constant (3 x 108 m/s), energy released is directly related to the mass defect • Therefore, to release more energy the mass defect must be increased, and vice versa • This formula shows that small masses can produce large amounts of energy as mass is multiplied by a huge number (speed of light squared)
Nuclear Fission I • In nuclear fission a ‘heavy’ nucleus splits into lighter nuclei • When a neutron is absorbed by an atom’s nucleus, it becomes unstable, causing it to split • For instance, when a neutron is absorbed by uranium 235, it turns into uranium 236, which then splits causing a reaction • atoms such as Barium-141 and Krypton-92 are the products of this “splitting” • During fission reactions, neutrons are produced • These neutrons can be used to collide with other nuclei • This results in more energy and neutrons, but more fission will be taking place This is called a chain reaction • A chain reaction means that since the neutrons created from one reaction can collide with other nuclei, the reaction is self- sustaining, as it can keep going by itself
Nuclear Fission II A chain reaction does not occur in every nuclear fission reaction • To have a chain reaction the mass of the original element has to be at the ‘critical mass’ • If it is less than the critical mass, then the neutron will escape and will not cause more reactions • The term critical mass is when there is no increase or decrease in the amount of neutrons emitted, temperature, and power. It is a state of equilibrium fission reaction • In production of electricity through nuclear power, a critical reaction is the ideal reaction needed as sub/super critical can overheat the machinery Splitting Atoms. Enteractive. 01 Feb 1995.
Nuclear Fission III • Fission can only start by a neutron and not by an alpha particle or an electron • The positive particle would be repelled by the nucleus • The electron would not be able to disturb the nucleus because it has too small of a mass • In nuclear fission the mass of the original atom is greater than that of the 2 smaller nuclei • This is because some of it was converted into energy • Scientist Lise Meitner found that the mass of the two nuclei formed added up were lighter than the nuclei of the original Uranium nucleus by approximately 1/5th of a proton. Hence, in this process, the “disappeared mass” was converted to energy given, in agreement with the formula E=mc2 • Nuclear fission was used on the bomb ‘Little Boy’ that was dropped by the US on Hiroshima, Japan during WWII
Nuclear Fusion I • Nuclear fusion is when two nuclei form together to produce a heavier one • Large temperatures are necessary for the fusion of light nuclei • This is because their is a natural electrostatic repulsion between the nuclei • As a result, by having high temperatures this repulsion is overpowered, and fusion can take place • Nuclear fusion is commonly done using forms of hydrogen, deuterium and tritium • Helium-4 is formed along with a free neutron as a result of this fusion Nuclear Fusion. Enteractive. 01 Feb 1995.
Nuclear Fusion II • Nuclear fusion occurs in stars and is the source of energy for stars • On stars due to the high temperature nuclear fusion can take place • During nuclear fusion, the product after the nuclei merge, has a mass slightly smaller than the mass of the 2 original nuclei • As with nuclear fission, the extra mass is converted into energy with agreement to E=mc2 • The hydrogen bomb is created through fusion • The first successful test of the hydrogen bomb was done by the US in November 1951 • Hydrogen nuclei fuse to create helium, and large amount of energy is released
Nuclear Fusion III • Nuclear fusion is not used as a source for commercial energy • This is because, since fusion occurs at very high temperatures, it is not yet possible for more energy to produced by fusion, then was originally put in to get the process going • Commercial energy comes from the fission process now • Nuclear fusion on the whole may have more of an advantage than nuclear fission • Nuclear fusion produces a lot more energy, and there is less problem with radioactive waste, as there are with nuclear fission • This is because nuclear fusion currently yields a higher % energy then nuclear fission
http://www.youtube.com/watch?v=N7C14UIKuv8 – Small video showing how nuclear fission occurs Video
Uranium Atom: U-235, U-238 What is it: • heaviest naturally occurring element • found in the Earth’s crust Just like all other naturally elements it is found in different forms; these forms are known as isotopes. Isotopes are when there are a different number of neutrons (n0) in the nucleus of an atom. →In the case of the Uranium element, it occurs in 16 possible isotopes however, it is found as a combination of U-238 which is 99.3% of the reserves in the crust and U-235 which is 0.7% of the reserves. Taylor, John J. "The Nuclear Power Bargain". Issues in Science and Technology 3(2004):41.
Uranium in Nuclear Energy • U-235 and U-238 are the more important isotopes → U-235 is an isotope that under favourable conditions, such as in a nuclear reactor can split providing a lot of energy known as nuclear energy through nuclear fission. → U-238 is also an isotope that provides energy by decaying. → has a very slow rate of decay which makes it highly non-radioactive →provides 0.1 watt of energy per tonne of U-238 which is enough to warm up the Earth`s crust. "Physics: Nuclear Physics". The New York Public Library Science Desk Reference. 01 Jan 1995.
How to Get Uranium • Uranium is a metal that occurs naturally in the Earth`s crust and is therefore found as an ore. → There are many areas on the surface of the Earth where the ore can be found, including in seawater → just as all metals that occur naturally, Uranium is also found in high concentrations in certain areas of the world. • deposits of Uranium ore are called an orebody. → these orebodies causes the industrial mining of the Uranium to be a lot more efficient. Australia has not only the largest orebody in the world but in effect, it also has the largest Nuclear Energy reactor. However, due to political reasons and participation, Canada is the largest Uranium supplier in the world.
How to Get Uranium • Uranium is only sold and exported to countries that are a part of the Nuclear Non-Proliferation Treaty • This ensures that the Uranium is used specifically for peaceful reasons and not for weapon development. Uranium Deposits "nuclear energy". Compton's by Britannica, v 6.0. 27 Jan 2009.
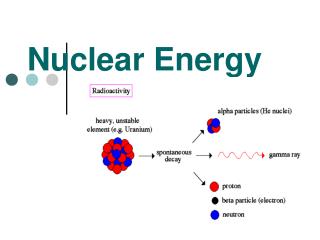
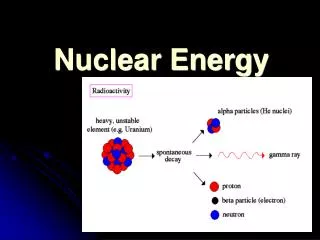
Radiation and Nuclear reactions • In 1902, Frederick Soddy proposed that radioactivity is a result of a natural change in atoms • This is a change of an isotope of one element into an isotope of another element • All elements heavier than Bismuth are naturally radioactive, thus they have the ability to decay into lighter elements • This is caused by a change in the number of protons, thus changing the element itself • The three common types of radiation and nuclear reactions are : Alpha, Beta and Gamma
Types of Radiation Alpha Radiation • This type of radiation is the result of the emission of an alpha particle from the nucleus • Each alpha particle consists of two protons and two neutrons • This means that the atomic number of the atom will decrease by two • This element has “transmuted” into another element that is exactly two atomic numbers lower • I.e. Uranium when it decays will transmute into Thorium which is two atomic numbers lower on the periodic table
Types of Radiation Beta Radiation Gamma Radiation • This type of radiation happens when a neutron is transmuted into a proton and an electron • When this occurs atomic mass does not change, because the neutron has simply turned into a proton • However the atomic number increases by one • This is because atomic number is determined by the number of protons • This type of radiation is caused by the emission of electromagnetic energy • Excited atoms may release photons, which are packets of electromagnetic energy • This allows the atom to move from an unstable state to a more stable state • This radiation does not involve any emissions or changes in particles, thus not transmutation occurs • However, as gamma radiation is released, it often triggers alpha and beta radiation
Nuclear Power plants • Nuclear Power-plants and Fossil-fuelled power stations have many aspects in common → both need steam to turn turbines and function generators BUT, in a Nuclear power-plant the fossil fuel burning is replaced by Uranium fission • The chain reaction which takes place inside the reactor is controlled by a set of rods → capable of absorbing the neutrons that are released by Nuclear Fission → rods can be pulled in or out of the compartment depending on the desired power level. • The power level may prove as a problem at times due to the high voltage that is being created by the plant BUT, a safety measure is that a plant will not explode but it will melt down therefore it does not spread as much radiation as expected and this also happens extremely rarely.
Nuclear Reactors • A Nuclear Reactor is a device where nuclear chain reactions are initiated, controlled and sustained. • it generates electrical power through a primary energy source. • The main purpose of the reactor is to convert nuclear energy into heat. IRAN-NUCLEAR-REACTOR. Agence France Presse. 22 Aug 2004.
Types of Nuclear Reactors Pressurized Water Reactors (PWR) • A pressure vessel is used in this reactor to hold the nuclear fuel, control rods, moderator, and coolant. • It is controlled by high pressure liquid water. • The pressure vessel releases radioactive water which is then looped through a steam generator, which heats another loop of water to steam which run turbines. • This is the most popular current reactors, and is considered the safest and most reliable nuclear technology.
Types of Nuclear Reactors Boiling Water Reactors(BWR) • similar to PWR, however they are without a steam generator. • Like a PWR, BWR is cooled and moderated by water; however it is at a lower pressure. This allows water to boil inside the pressure vessel which produces steam and in turn runs the turbines. • no primary and secondary loops are in PWR. • Are potentially more stable and safe because the thermal efficiency can be higher, and therefore simpler.
Types of Nuclear Reactors Pressurized Heavy Water Reactor(PHWR) • This is a Canadian design (also known as CANDU), these reactors are a combination of heavy-water-cooled and moderated versions of Pressurized-Water reactors. • unlike the PWR, a single large pressure vessel is replaced with hundreds of smaller pressure tubes. • fueled with natural uranium and have the design of a thermal neutron reactor. • Although a Canadian invention, these reactors show up in numerous parts of Asia, South America and Europe.
Types of Nuclear Reactors ReaktorBolshoyMoshchnostiKanalniy (High Power Channel Reactor) (RBMK) • This Soviet Union design was built not only to produce power, but as well plutonium. • Uses cool water and a graphite moderator. Similar to CANDU because • both are re-fuelable during power operation, • use a pressure tube design rather than a pressure vessel like the ones used in PWRs Different to CANDU because • RBMKs are very unstable, large which in turn makes them dangerous in the case of an accident. Fact: This is the same reactor that causes the tragic disaster that took place in the Chernobyl plant.
Types of Nuclear Reactors Gas Cooled Reactor (GCR) and Advanced Gas Cooled Reactor(AGCR) • Usually graphite moderated and CO2cooled. • Due to their higher operating temperatures, they have a higher thermal efficiency. Many of these reactors are found in the United Kingdom. • AGCRs have a predicted life of 10 to 20 years.
Types of Nuclear Reactors Liquid Metal Fast Breeder Reactor (LMFBR) • cooled by liquid metal and completely unmoderated, • produces more fuel than it consumes. • called “Breeder Reactors” because they produce fissionable fuel as a result of neutron capture during operation. • function similar to PWR efficiency wise, and high pressure containment isn’t required as the liquid metal doesn’t need to be kept at a high pressure, even at very high temperatures.
Safety: Maintaining a Nuclear Reactor Control of Radioactivity • Radioactivity causes many harmful effects to the body • It is important that the neutron flux can be controlled. • In a nuclear reactor when a neutron is captured by a fuel nucleus (usually uranium) the nucleus of the atom is split and in turn released radioactive particles (or undergoes fission). • Therefore, decrease in neutron flux=decrease in radioactivity • A way to reduce neutron flux: • include neutron-absorbing control rods partially inserted into the reactor core which will then reduce the reactions. • control rods are an essential component as the reaction could run out of control if fission events are extremely frequent.
Safety: Maintaining a Nuclear Reactor Maintenance of Core Cooling • It is essential that there is cooling of some sort in every nuclear reactor. (to prevent overheating etc.) • The most common cooling agent used in nuclear reactors is water, although some do use sodium or sodium salts. Maintenance of barriers that prevent the release of radiation • physical barriers are between the radioactive core and the environment. • this shields the workers in nuclear power plants from radiation by concrete walls within the plant. • Next a vacuum building, a 71m high concrete structure, is connected to reactor buildings which are kept at negative atmospheric pressure. • The purpose of the vacuum building is that if by any chance radiation were to be leaked from the reactor, the vacuum would suck it in and in turn prevent it from being leaked into the environment. It is essential that the design of reactors include multiple back-up components (two or more independent systems performing the same function in parallel), to prevent failure of any equipment.
The Safety of the Workers There are also rules and regulations workers of nuclear power plants must follow in order to ensure their safety. • The handling of equipment solely by remote in the core of the reactor • Physical shielding is required • Time restriction on the time a worker spends in areas with significant radiation levels • Monitoring of individual doses and of the work environment http://www.world-nuclear.org/education/graphics/wast1.gif
Accidents that have occurred Three Mile Island (Wednesday, March 28, 1979 Three Mile Island, USA. ) • second most severe accident to have happen due to Nuclear Power Plants. • Caused by a failure in the secondary system, which was followed by the opening of a pilot-operated relief valve (PORV) in the primary part of the system. • This led to significant amounts of reactor coolant to escape. • The reactor was fortunately brought under control and there weren’t any casualties or long term affects due to radiation. http://www.ohiocitizen.org/campaigns/electric/2004/ph_three_mile_island500.jpg
Accidents that have occurred Chernobyl (April 26, 1986 Chernobyl Nuclear Power Plant in Ukraine) • the largest and most tragic accident to have ever occurred in civil nuclear history. • The result of this was a mass release of radioactivity as well power excursion which destroyed the reactor. • The fire sent a plume of highly radioactive fallout over large parts of the western Soviet Union, Eastern Europe, Western Europe, Northern Europe, and as far as Ireland. • This led to the resettlement of over 336,000 people. • According to statistics, 56 people died directly from the accident, of which 47 workers and nine children diagnosed with thyroid cancer. • It was also estimated that an extra 4,000 were killed due to cancer, and approximately 600,000 people were exposed to the high radioactive substance.
Nuclear Waste • The world's major military powers have built tens of thousands of atomic bombs powerful enough to kill the world's inhabitants several times over, and, despite the fact that nuclear disarmament is occurring between the super powers, it is a very slow process. • Tons of radioactive waste is produced each year from reactors, some of which has the potential to stay dangerous for over 5,000,000 years • Once this is created, many of the nuclear by-products stay in our biosphere hundreds of thousands of years, which does irreversible damage to not only the plant, but animals and human life. • Technology is advancing everyday to very significant extents, and this would include nuclear weapons as an unfortunate possibility in the future. • The energy input essential to build and operate nuclear plants as well mine and process the uranium is so large and causes a net energy deficit. The input energy is 4.8 percent of output energy when diffusion is utilized to enrich uranium and 1.7 percent if newer technology is used.
Mutations • It takes just one radioactive atom, one cell, and one gene to initiate the cancer or mutation cycle. Therefore, an exposure can pose multiple threats to a human’s life. • The increase of radiation exposure to the workers, the general public and environment around can be tragic for humans. This could imply an increase in number of people who will deal with cancer or offsprings possessing deformities or disease as humans are very susceptible and sensitive to radiation. • Nuclear radiation damages cells all over the body making people sick or even killing them, and illnesses can strike many years after.
Chromosomes Human Chromosome damaged by radiation Normal human Chromosome Source: www.cancerquest.org Source: www.cancerquest.org
Table 1: below is a table that shows the dose of radiation (in unit rem) that is required to cause a certain degree of damage
Timeline 1930s: Significant discoveries are made about the FISSION OF ATOMS by Enrico Fermi, Otto Hahn, Fritz Strassmann and Lise Meitner. December 2, 1942: Enrico Fermi achieves a controlled NUCLEAR CHAIN REACTION with a demonstration reactor, called the Chicago Pile 1 August 6, 1945: United States drops an ATOMIC BOMB on Hiroshima, then on August 9th on Nagasaki October 6, 1947: US Atomic Energy Commission looks into possibly using atomic energy for PEACEFUL USES December 20, 1951: Experimental REACTOR PRODUCES ENERGY from a nuclear reaction for the first time, enough to light four light bulbs January, 1955: the Atomic Energy Commission begins program of FUNDING for nuclear power plants between government and industry 1956: The FIRST NUCLEAR POWER STATION was built. Using uranium as its fuel. The station was named Calder Hall Power Station, built on coast of Cumberland December 2, 1957: In Shippingport, Pennsylvania, the first full scale NUCLEAR POWER PLANT goes into service.
Timeline Continued April 3, 1965: The FIRST NUCLEAR REACTOR is operated from outer space 1973: American utilities buy 41 NUCLEAR POWER PLANTS March 28, 1979: at Three Mile Island near Harrisburg, Pennsylvania a major NUCLEAR ACCIDENT occurs. Fortunately no one is hurt, but all damage is to the reactor of the plant January, 1983: President Reagan signs the NUCLEAR WASTE POLICY ACT 1984: NUCLEAR POWER SURPASSES Hydro power in total electrical generation in 1984. April 26, 1986: In the unit 4 reactor of the Chernobyl Nuclear plant were two EXPLOSIONS. This disaster exposed millions of people to radioactive isotopes. Been linked many forms of cancer in natives of eastern Europe and Russia, as well as destroying animals and plants December 1993: 109 NUCLEAR POWER PLANTS in total in the United States, producing 610 billion kWhs of electricity.
Uses of Nuclear Energy • Other uses of Nuclear energy are dependent on radio isotopes which are isotopes that are highly radioactive with the natural environment of the Earth and over the past few decades, radio isotopes have drastically changed the lives of the layman. • Radio isotopes are artificially produced as Uranium does not exist as a radioisotope at all or often enough for it to be used. • The production of these isotopes is both fast and cost efficient as provide many different uses in the day-to-day life. "NUCLEAR ENERGY". Young Students Learning Library. 01 Jan 1996.
Benefits of Radioisotopes • Food Preservation: The use of radioisotopes in order to preserve foods and in order to kill parasites and other germs after harvesting is becoming more and more popular over the years. • Crops and Livestock: Farmers use radioisotopes to make fertilizers • Medicine: Radio isotopes provide a vast amount of aid in the medical world. Radioisotopes are used for diagnostic purposes. E.g. X-rays, and the treatment of cancer "Physical Sciences and Mathematics: nuclear reaction". The Dictionary of Cultural Literacy. 01 Jan 1988.
Increasing Demand for Nuclear Energy As the processes by which Nuclear Energy is obtained is becoming more popular, the means by which electricity is being produced throughout the world is changing. Nuclear energy is becoming more and more popular meaning that there are less fossil fuel that are being exhausted and in turn there is less damage being done to the environment therefore increasing the biological health of the Earth making it a safer place. Source: Energy Information Administration International Energy Annual 2004 Updated 2007
Nuclear Energy Today • Nuclear energy which is fast, effective and Earth friendly has grown in popularity → has given it a place among one of the top four ways of producing electricity across the globe. HOWEVER, energy produced by fossil fuel are still the largest source of energy BUT, Nuclear energy will soon be one of the top three contributors of energy. According to stats, USA is already 1/3 of the nuclear activity • There has been a lot of progress in the development of Nuclear Reactors → Over the past decade, the number of nuclear reactors has not increased much BUT, what’s increased is the amount of energy that is produced by these reactors. The average load on one reactor has shot up to 90% which was 66% in 1990 and 56% in 1980
World Energy Usage During 2007 Table 2: . “Nuclear Electricity Generation 2007”. World Nuclear Association. 09 Jan 2007.
Social Benefits to Nuclear Energy • The growth and development of Third World countries poses the threat of an energy crisis. Currently the Third World population is expected to double to 8 billion people by mid-century. • The amount of energy used within these Third World countries is expected to increase as their infrastructure improves To supply this energy demand here is why Nuclear Energy is most viable: Table 3:
Thus Nuclear Energy Will: • Prevent Economic stagnation by encouraging the growth of Third World Countries-knowing future energy demands will be met • Resolve Energy Conflicts because nuclear energy means adequate supplies-no more competition over oil rigs • Compared to our current methods of obtaining energy, Nuclear Energy prevents environmental degradation Dilemma of Nuclear Power (Part 1 of 2). Enteractive. 01 Feb 1995.
Risks of Nuclear Energy RISKS As with any type of Energy, Nuclear energy has its share of disadvantages and risks which include: • The product of the process is a great deal of radioactive NUCLEAR WASTE that is currently being stored underground. • The fission reaction needs to be very controlled as it has a LARGE POSSIBLE RISK should anything go wrong. • Nuclear energy is a NONRENEWABLE energy source and so it can not be reused again once it has gone through the process. • The COST OF MAINTENANCE is very high as worn out nuclear power plants must be decommissioned, a massive undertaking that is very costly. In addition, accident insurance and safe waste disposal add to the high list of expenses. • When mining for nuclear fuel such as uranium, a great amount of CO2 IS RELEASED, causing hazardous waste which is harmful to plants and animals. • This can be a health concern as the RADIATION from nuclear fission can results in physical, mental and genetic changes. These changes can be passed on to children, resulting in congenital defects and disorders.
The Main Ingredient in Weapons of Mass Destruction? Nuclear bomb vs. Nuclear Power Plant In a Nuclear bomb, rapid reactions between atoms are occurring. In a Nuclear Power Plant, the speed of that reactions is controlled to create a supply of heat that can be used to produce steam and drive electric generations.
The Survey 1a. Do you know what nuclear energy is? Yes No 1b. If yes, do you believe there's more positive or negative feedback on this type of energy? Negative Positive Equal Neither 2. The production of nuclear energy is clean however the process required to obtain the uranium for nuclear fission is not. Relative to other sources of energy, do you believe nuclear energy is clean? Yes I believe nuclear energy is clean. No I do not believe nuclear energy is clean. 3. Put the following sources of energy in order from most clean to less clean. ______ Geothermal ______ Solar ______ Wind ______ Hydro ______ Biomass ______ Natural gas ______ Oil ______ Coal ______ Nuclear 4. According to Albert Einstein "The discovery of nuclear reactions need not bring about the destruction of mankind any more than the discovery of matches." Do you agree or disagree with this statement in reference to the effect that nuclear weapons have left on humanity. I Agree I Disagree 5. Do you support this source of energy? Yes, I support nuclear energy No, I don’t support nuclear energy
What does the Public have to say? Table 5: Table 4: Survey Tally