Basic Biochemistry
Basic Biochemistry. What is Biochemistry?. Biochemistry is the study of the chemical interactions of living things. Biochemists study the structures and physical properties of biological molecules. Often are involved in the manufacture of new drugs and medical treatments. CHEMISTRY OF LIFE.


Basic Biochemistry
E N D
Presentation Transcript
Basic Biochemistry KRT-2011
What is Biochemistry? • Biochemistry is the study of the chemical interactions of living things. • Biochemists study the structures and physical properties of biological molecules. • Often are involved in the manufacture of new drugs and medical treatments KRT-2011
CHEMISTRY OF LIFE • Elements: simplest form of a substance - cannot be broken down any further without changing what it is • Atom: the actual basic unit - composed of protons, neutrons, and electrons KRT-2011
THE ATOM • Just like cells are the basic unit of life, the ATOM is the basic unit of matter. • They are very small. If placed side by side one million would stretch a distance of 1cm. • The atom is made up of 3 particles. KRT-2011
Electrons are not present within the atom, instead THEY REVOLVE AROUND THE NUCELUS OF THE ATOM & FORM THE ELECTRON CLOUD • Draw a helium atom. Indicate where the protons, neutrons and electrons are. NEUTRONS PROTONS ATOMIC # = 2 (PROTONS) ATOMIC MASS = 4 (PROTONS & NEUTRONS) - + + ELECTRONS - KRT-2011
ISOTOPES • atoms of the same element that HAVE A DIFFERENT NUMBER OF NEUTRONS • Some isotopes are radioactive. This means that their nuclei is unstable and will break down at a CONSTANT RATE over time. • There are several practical uses for radioactive isotopes: • CARBON DATING • TRACERS • KILL BACTERIA / CANCER CELLS KRT-2011
COMPOUNDS • a substance formed by the chemical combination of 2 or more elements in definite proportions • Ex: water, salt, glucose, carbon dioxide H20 CO2 NaCl KRT-2011
The cell is a COMPLEX CHEMICAL FACTORY containing some of the same elements found in the nonliving environment. • carbon (C), hydrogen (H), oxygen (O), and nitrogen (N) are present in the greatest percentages KRT-2011
TWO TYPES OF COMPOUNDS • Organic - Contain C, H, and O in some ratio (usually referred to as chemicals of life) • Carbohydrates, Proteins, Lipids, Nucleic Acids • Inorganic - usually "support" life - no specific ratio of C, H, and O • Water (H2O), Carbon Dioxide (CO2) KRT-2011
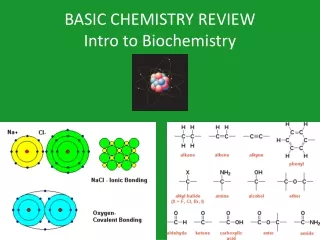
CHEMICAL BONDS • Chemical bonds hold the atoms in a molecule together. • There are 2 types of chemical bonds IONIC and COVALENT KRT-2011
IONIC BONDS • Occur when 1 or more electrons are TRANSFERRED from one atom to another. • When an atom loses an electron it is a POSITIVE charge. • When an atom gains an electron it is a NEGATIVE charge • These newly charged atoms are now called IONS • Example: NaCl (SALT) KRT-2011
COVALENT BONDS • Occur when electrons are SHARED by atoms. • These new structures that result from covalent bonds are called MOLECULES • ** In general, the more chemical bonds a molecule has the more energy it contains SHARING IS CARING! KRT-2011
MIXTURES • Water is not always pure. It is often found as part of a mixture. • A mixture is a material composed of TWO OR MORE ELEMENTS OR COMPOUNDS THAT ARE PHYSICALLY MIXED • Ex: salt & pepper mixed, sugar and sand – can be easily separated KRT-2011
SOLUTION Two parts: • SOLUTE – SUBSTANCE THAT IS BEING DISSOLVED (SUGAR / SALT) • SOLVENT - the substance in which the solute dissolves • Materials that do not dissolve are known as SUSPENSIONS. • Blood is the most common example of a suspension. • Cells & other particles remain in suspension. KRT-2011
FORMULA • The chemical symbols and numbers that compose a compound ("recipe") • Structural Formula – Line drawings of the compound that shows the elements in proportion and how they are bonded • Molecular Formula – the ACTUAL formula for a compound C2H6O KRT-2011
ACIDS & BASES • Acids: always (almost) begin with "H" because of the excess of H+ ions (hydrogen) • Ex: lemon juice (6), stomach acid (1.5), acid rain (4.5), normal rain (6) Facts about Acids • Acids turn litmus paper BLUE and usually taste SOUR. • You eat acids daily (coffee, vinegar, soda, spicy foods, etc…) KRT-2011
ACIDS & BASES • Bases: always (almost) end with -OH because of the excess of hydroxide ions (Oxygen & Hydrogen) • EX: oven cleaner, bleach, ammonia, sea water, blood, pure water Facts about Bases • Bases turn litmus BLUE. • Bases usually feel SLIPPERY to touch and taste BITTER. KRT-2011
Neutralization Reactions • When an acid reacts with a base to produce a salt and water. KRT-2011
pH SCALE • measures degree of substance alkalinity or acidity • Ranges from 0 to 14 • 0 – 5 strong acid • 6-7 neutral • 8-14 strong base KRT-2011
The goal of the body is to maintain HOMEOSTASIS (neutrality) – to do this when pH is concerned, we add weak acids & bases to prevent sharp changes in pH. • These are called BUFFERS KRT-2011
The most common elements found in living organisms include: Carbon (C) Oxygen (O) Nitrogen (N) Hydrogen (H) Phosphorus (P) Sulfur (S) Elements in Living Organisms KRT-2011
Biochemistry: where chemistry and biology meet head-on • Living things require millions of chemical reactions within the body, just to survive. • Metabolism = all the chemical reactions occurring in the body. • Organic molecules: • usually associated with living things. • always contain CARBON. • are “large” molecules, with many atoms • always have covalent bonds (share electrons) KRT-2011
Plant Metabolism KRT-2011
Primary Metabolites • Primary metabolites are compounds that are commonly produced by all plants and that are directly used in plant growth and development. • The main primary metabolites are carbohydrates,lipids, proteins, and nucleic acids. KRT-2011
ORGANIC COMPOUNDS LIPIDS PROTEINS NUCLEIC ACIDS CARBOHYDRATES KRT-2011
Macromolecules of Cells • Macro = large • 4 types of macromolecules in cellular biology 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids (DNA & RNA) KRT-2011
Macromolecule #1: Carbohydrates • Sugars and groups of sugars • Purposes: energy and structure • Includes three types: • Monosaccharide (1 sugar – quick energy) • Disaccharide (2 sugars – short storage) • Polysaccharide (many sugars – energy long storage & form structures) KRT-2011
Carbohydrates • Carbohydrates are the sugars made up of glucose and its isomers • Carbohydrates come in many different sizes: • Monosaccharides made up of one sugar unit (glucose or fructose) • Disaccharides made up of two sugar units (sucrose is a glucose and a fructose) • Polysaccharides are polymers made up of more than two sugar units KRT-2011
Monosaccharides (simple sugars) • all have the formula C6 H12 O6 • all have a single ring structure • (glucose is an example) KRT-2011
Disaccharides (double sugars) • all have the formula C12 H22 O11 • sucrose (table sugar) is an example KRT-2011
Harvesting Sucrose Sugar Cane Maple Syrup KRT-2011
Refining Sucrose KRT-2011
Macromolecule #1: Carbohydrates • Polysaccharide Examples: • Glycogen—glucose polymer stored for future energy needs. Found in liver, muscle and sperm, etc. • Cellulose—glucose polymer used to form fibers for plant structures. Humans can’t digest (fiber). Most abundant organic molecule. • Chitin—glucose polymer for exoskeletons of some crustaceans & insects. KRT-2011
Polysaccharides • Structural polysaccharides are used to support plants • Storage polysaccharides are used to store energy for later use by the plant KRT-2011
Structural Polysaccharides • The most common structural polysaccharide in plants is cellulose. It makes up 40 to 60% of the cell wall. It is also the most common polymer on earth • Cellulose is extremely strong due to its chemical organization. It is made of a long chain of beta-glucose molecules – 100 to 15,000 glucose molecules KRT-2011
CARBOHYDRATES • Living things use carbohydrates as a key source of ENERGY! • Plants use carbohydrates for structure (CELLULOSE) • include sugars and complex carbohydrates (starches) • contain the elements carbon, hydrogen, and oxygen (the hydrogen is in a 2:1 ratio to oxygen) KRT-2011
Polysaccharides • Formed of three or more simple sugar units • Glycogen - animal starch stored in liver & muscles • Cellulose - indigestible in humans - forms cell walls • Starches - used as energy storage KRT-2011
Cotton Boll – Pure Cellulose KRT-2011
Polysaccharides KRT-2011
Polysaccharides KRT-2011
Gluey Polysaccharides • Pectins are mainly polymers of galacturonic acid. • Hemicelluloses are highly variable and are not related to cellulose. • Grass hemicelluloses are high in xylose, with small amounts of arabinose, galactose, and urionic acids. But pea family (Fabaceae) are high in arabinose, galactose and urionic acid, but low in xylose. • Some of the most interesting hemicelluloses are not actually used structurally, but rather are exuded from stems, leaves, roots, or fruits in a sticky mixture called a gum KRT-2011
Pectin and Hemicellulose KRT-2011
Gum Arabic from Acacia senegal KRT-2011
Storage Polysaccharides • The most important storage polysaccharides are amylose and amylopectin. Amylose is a long chain of alpha-glucose, several hundred to several thousand molecules long. Amylopectin is more complex, often made up of 50,000 molecules. • These two polymers are both used in making starch grains. Most starch grains are about 20% amylose and 80% amylopectin, but this varies with the plant. KRT-2011