Balancing Chemical Equations: Steps, Solutions & Practice
140 likes | 658 Vues
Learn how to balance chemical equations step by step, check atom balances, and practice with solutions for various reactions. Improve your understanding of balancing equations in chemistry.

Balancing Chemical Equations: Steps, Solutions & Practice
E N D
Presentation Transcript
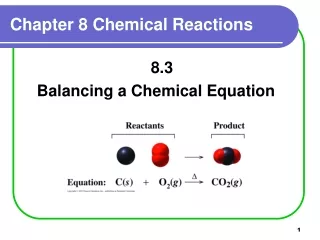
Chapter 8 Chemical Reactions 8.3 Balancing a Chemical Equation
Balancing Chemical Equations 1. Write the equation with the correct formulas. NH3(g) + O2(g) NO(g) + H2O(g) 2. Determine if the equation is balanced. No, not all atoms are balanced. 3. Balance with coefficients in front of formulas. 4NH3 + 5O24NO + 6H2O 4. Check that atoms of each element are equal in reactants and products. 4 N (4 x 1N) = 4 N (4 x 1N) 12 H (4 x 3H) = 12 H (6 x 2H) 10 O (5 x 2O) = 10 O (4O + 6O)
Steps in Balancing an Equation To balance the following equation, Fe3O4(s) + H2(g) Fe(s) +H2O(l) • Work on one element at a time. • Use only coefficients in front of formulas. • Do not change any subscripts. Fe: Fe3O4(s) + H2(g) 3Fe(s) +H2O(l) O:Fe3O4(s) + H2(g) 3Fe(s) +4H2O(l) H: Fe3O4(s) + 4H2(g) 3Fe(s) +4H2O(l)
Learning Check Check the balance of atoms in the following: Fe3O4(s) + 4H2(g) 3Fe(s) +4H2O(l) A. Number of H atoms in products. 1) 2 2) 4 3) 8 B. Number of O atoms in reactants. 1) 2 2) 4 3) 8 C. Number of Fe atoms in reactants. 1) 1 2) 3 3) 4
Solution Fe3O4(s) + 4H2(g) 3Fe(s) +4H2O(l) A. Number of H atoms in products. 3) 8 (4H2O) B. Number of O atoms in reactants. 2) 4 (Fe3O4) C. Number of Fe atoms in reactants. 2) 3 (Fe3O4)
Checking a Balanced Equation Reactants Products 1 C atom = 1 C atom 4 H atoms = 4 H atoms 4 O atoms = 4 O atoms
Learning Check Balance each equation and list the coefficients in the balanced equation going from reactants to products: A. __Mg(s) + __N2(g) __Mg3N2(s) 1) 1, 3, 2 2) 3, 1, 2 3) 3, 1, 1 B. __Al(s) + __Cl2(g) __AlCl3(s) 1) 3, 3, 2 2) 1, 3, 1 3) 2, 3, 2
Solution A. 3) 3, 1, 1 3Mg(s) + 1N2(g) 1Mg3N2(s) B. 3) 2, 3, 2 2Al(s) + 3Cl2(g) 2AlCl3(s)
Balancing with Polyatomic Ions MgCl2(aq) + Na3PO4(aq) NaCl(aq) + Mg3(PO4)2(s) • Balance PO43-as a unit. MgCl2(aq) + 2Na3PO4(aq) NaCl(aq) + Mg3(PO4)2(s) 2 PO43- = 2 PO43- • Balance Mg and Cl. 3MgCl2(aq) + 2Na3PO4(aq) 6NaCl(aq) + Mg3(PO4)2(s) 3 Mg2+ = 3 Mg2+ 6 Na+ = 6 Na+ 6 Cl- = 6 Cl-
Learning Check Balance and list the coefficients from reactants to products: A. __Fe2O3(s) + __C(s) __Fe(s) + __CO2(g) 1) 2, 3, 2,3 2) 2, 3, 4, 3 3) 1, 1, 2, 3 B. __Al(s) + __FeO(s) __Fe(s) + __Al2O3(s) 1) 2, 3, 3, 1 2) 2, 1, 1, 1 3) 3, 3, 3, 1 C. __Al(s) + __H2SO4(aq) __Al2(SO4)3(aq) + __H2(g) 1) 3, 2, 1, 22) 2, 3, 1, 3 3) 2, 3, 2, 3
Solution A. 2) 2, 3, 4, 3 2Fe2O3(s)+ 3C(s) 4Fe(s) + 3CO2(g) B. 1) 2, 3, 3, 1 2Al(s) + 3FeO(s) 3Fe(s) + 1Al2O3(s) C. 2) 2, 3, 1, 3 2Al(s) + 3H2SO4(aq) 1Al2(SO4)3(aq) + 3H2(g)