Weak Acids
This study explores the behavior of weak acids, specifically vinegar, highlighting the two forms of acetic acid in solution—free acid and conjugate base. The dissociation constants (Ka and pKa) illustrate how poorly weak acids dissociate, affecting their pH behavior. Through titration with NaOH, the relationship between the percentage of acid and base is examined, underlining the Henderson-Hasselbalch equation's role in predicting acid-base ratios. The unique characteristics of vinegar are analyzed, including its smell and membrane permeability in relation to its pH changes during titration.

Weak Acids
E N D
Presentation Transcript
Weak Acids Acidity and Behavior, the Vinegar Example
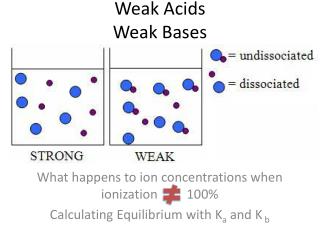
Weak Acids Dissociate Poorly as Seen by Their Dissociation Constants, Ka In other words, only one in a hundred protons dissociates!
And Its -log10, pKa • pKa = -log10Ka • Ka for acetic acid is 1.76 x 10-5M, • pKa = -log 1.76 x 10-5= ? • pKa for formic acid is 3.75, • Ka = 10-pKa= 10-3.75 = ?
Which Form Will Be Present? • Free acid form occurs at low pH • Conjugate base occurs at high pH • At some point amount of acid equals amount of base
Predicts the Ratio of Acid and Base, • What is the ratio of Ac- to HAc at pH 2.8? (See example on the right) • Practice: What is the ratio of Ac- to HAc at pH 6.8?
but Not the pH • pKa is the pH where there are equal amounts of free acid and conjugate base • pH is dependent on the concentration of acid as well as its pKa • pKa ≠ pH of a solution, pH is usually <pKa The above formula is derived from the equilibrium constant equation assuming [H+] = [Ac-], and [HAc] >> [Ac-] The lower the pKa, the less valid the last assumption is.
What’s going on at pKa? OrWhen does [HAc] = [Ac-]? When pH = pKa you can simplify the notation by representing the ratio with r, replace it in the equation, and solve its value. The ratio is 1 only when
Landmarks on the titration curve Mostly Ac- Present [HAc] = [Ac-] pKa Mostly HAc Present
Behavior of Vinegar Does not smell, can’t cross membranes pH of solution barely changes when base added Strong smell, crosses membranes