Weak Acids and Bases
220 likes | 369 Vues
Weak Acids and Bases. Mg (s) + 2HCl (aq) --> MgCl 2 (aq) + H 2 (aq) Mg (s) + 2CH 3 COOH (aq) --> Mg(CH 3 COO 2 ) 2 (aq) + H 2 (aq). Working with Weak Acids and Bases. In a solution of a weak acid, what do we have? Acid molecules Small amount of H 3 O +

Weak Acids and Bases
E N D
Presentation Transcript
Weak Acids and Bases Mg (s) + 2HCl (aq) --> MgCl2 (aq) + H2 (aq) Mg (s) + 2CH3COOH (aq) --> Mg(CH3COO2)2 (aq) + H2 (aq)
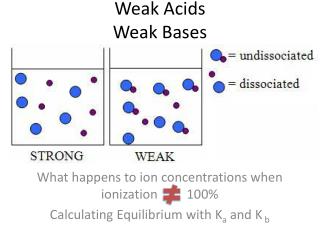
Working with Weak Acids and Bases • In a solution of a weak acid, what do we have? • Acid molecules • Small amount of H3O+ • Even smaller amount of OH- (to maintain autoprotolysis equilibrium) • Conjugate base • We see similar things for a solution of a weak base
Working with Weak Acids Let’s look at Acetic acid: CH3COOH (aq) + H2O (l) H3O+ (aq) + CH3CO2- (aq)
The Acid Dissociation Constant • A K value for the equilibrium reaction of an acid and water • Gives us an idea of how strong the acid is
The Base Ionization/Dissociation Constant • For a weak base, we can set up a similar calculation: NH3 (aq) + H2O (l) NH4+ (aq) + OH- (aq)
Conjugate Acids/Bases and Kw • We can write the Ka expression for an acid and the Kb for the conjugate base (or vice versa) NH4+ (aq) + H2O (l) H3O+ (aq) + NH3 (aq) NH3 (aq) + H2O (l) NH4+ (aq) + OH- (aq) Kw = Kax Kb pKw = pKa + pKb
Summary: Ionization/Dissociation Constants • The proton donating strength of an acid is measured by its dissociation constant, Ka • The proton accepting strength of a base is measured by its dissociation constant, Kb • The smaller the constant, the weaker the acid or base. The negative log of Ka or Kb is the pKa or pKb. • The higher the pK value, the weaker the acid or base • The stronger the acid or base, the weaker its conjugate base or acid will be.
Molecular Structure and Acid Strength • The strength of an acid depends on how easily the H-A bond is broken • We’ll use our previous studies of periodic trends and electronegativities to determine the strength of various acids
Binary Acid Strength • A binary acid is composed of hydrogen and one other element • H2O, H2S, HCl • In a binary acid, the greater the electronegativity of element A, the stronger the acid HA • Why? • Going down a group, the acid strengths increase • Why?
Oxoacid and Carboxylic Acid Strengths • The O-H bond is very polar which is one reason why the proton can leave so easily • Looking at hypohalous acids, what can we learn? HClO H-O-Cl HBrO H-O-Br HIO H-O-I Increasing acid strength. Why? Electronegative halogen ‘hogs’ electrons from oxygen which means that oxygen must, in turn, steal from hydrogen
Oxoacid Families and Acid Strength • As the number of oxygen atoms increases within the family, the acid strength increases
Carboxylic Acids (Organic Acids) • The more oxygens or electronegative atoms attached to a given carbon, the more acidic the proton or acid group
Chapter 11: Sections 1 through 5 • In this chapter, we are going to build upon the basics we learned in Chapters 9 and 10 to study the complex equilibria that exist in solutions • These techniques can be used in Qualitative Analysis, the study of the identification of substances present in a solution
Buffers • If we dissolve a weak acid or base in solution and then add some of a salt containing the conjugate base or acid, we have created a buffer. • A buffer is a mixed solution that resists a change in pH when a strong acid or base is added • Buffers are used in all branches of scientific endeavour and you yourself have a buffer system that regulates your blood pH.
Buffers: How They Work • Looking at acetic acid: CH3COOH (aq) + H2O (l) H3O+ (aq) + CH3COO- (aq) The pKa for this reaction is 4.75, the Ka is 1.78x10-5 (in other words, its ain’t happening THAT much) • If we added a few drops of strong acid to this solution, the pH would drop very sharply. Why? However, if we added an equivalent amount of NaCH3COO to the solution first, there would be an abundance of CH3COO- ions present These CH3COO- ions would then “absorb” the protons from any acids added and the pH wouldn’t change much
Buffers: How they work • We can make a similar basic buffer using aqueous ammonia and ammonium chloride NH3 (aq) + H2O (l) NH4+ (aq) + OH- (aq) NH4Cl (aq) + H2O (aq) NH4+ (aq) + Cl- (aq) + H2O (l) Added protons will react with ammonia to prevent the pH from decreasing Added Hydroxide will react with ammonium to prevent the pH from rising
A buffer is a mixture of a weak conjugate acid-base pair that stabilizes the pH of a solution by providing a source and a sink for protons.
Designing a Buffer • Identify the weak acid and its conjugate base • Setup the Ka equation and solve for [H3O+] • Calculate the pH by approximating the equilibrium concentrations of the acid and its conjugate base • They are essentially the same as the initial concentrations
Preparing a Buffer (Practical) If we have the same amount of weak acid and conjugate base (HA and A-) then [HA] = [A-] And: Therefore, when [HA]=[A-], the pKa=pH To make a buffer at a specific pH, find an acid with the pKa close to the desired pH and add an equimolar amount of its conjugate base
Henderson-Haselbach Equation We can use this equation to calculate the pH of a buffer solution directly All we need to know is [A-], [HA] and the Ka for the acid