Weak Acids Weak Bases
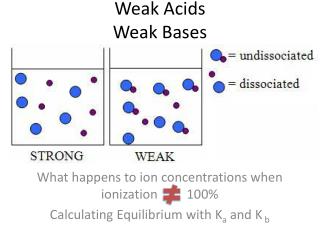
Weak Acids Weak Bases. What happens to ion concentrations when ionization 100% Calculating Equilibrium with K a and K b. VIDEO REVIEW. View Acid Ionization Video Clip View Base Ionization Video Clip. How do weak acids and weak bases differ from strong?. Weak Acids. K a =.


Weak Acids Weak Bases
E N D
Presentation Transcript
Weak AcidsWeak Bases What happens to ion concentrations when ionization 100% Calculating Equilibrium with Ka and K b
VIDEO REVIEW • View Acid Ionization Video Clip • View Base Ionization Video Clip How do weak acids and weak bases differ from strong?
Ka= [H3O+] [A-] [HA] HA (aq) + H2O (l) A-(aq) + H3O+(aq) Weak or Partial Ionization Expression This equilibrium constant is called the acid-dissociation constant, Ka. Turn to p. 667, Table 16.2 ALSO in APPENDIX D
Cause of Acidic Behavior • Read p. 667- Last Sentence
Relevance + Magnitude of Ka The greater the value of Ka, the stronger is the acid. Kavalues for weak acids range from 10-2to 10-10.
[H3O+] [COO-] [HCOOH] Ka = Sample Exercise 16.10 p. 668 The pH of a 0.10 Msolution of formic acid, HCOOH, at 25C is 2.38. Calculate Ka for formic acid at this temperature. We know that To calculate Ka, we need the equilibrium concentrations of all three things.
1. Use given pH to find [H+] pH = -log [H+] 2.38 = -log [H+] -2.38 = log [H+] 10-2.38 = 10log [H+] = [H+] 4.2 10-3 = [H+]
[4.2 10-3] [4.2 10-3] [0.10] Ka = 2. Set up ICE Chart 3. PLUG into Ka expression = 1.8 10-4
[H+]eq [HA]initial 4.2 10-3 0.10 Percent Ionization • Another measure of acid strength • The stronger the acid, the greater the percentage ionization • Percent Ionization = 100 • From previous example: [H+]eq = 4.2 10-3 M [HCOOH]initial~ 0.10 M 100 = 4.2%
Using Ka to calculate pH • Sample Exercise 16.12 p. 671
Weak Bases Bases react with water to produce hydroxide ion.
[HB] [OH-] [B-] Kb = Base Equilibrium Expression where Kb is the base-dissociation constant. Kb can be used to find [OH-] and, through it, pH.
NH3(aq) + H2O (l) NH4+ (aq) + OH-(aq) [NH4+] [OH-] [NH3] = 1.8 10-5 Kb = Sample Exercise 16. What is the pH of a 0.15 M solution of NH3?
= 1.8 10-5 = [NH4+] [OH-] [NH3] Kb = (x)2 (0.15) 2. Use ICE method to tabulate data. (1.8 10-5) (0.15) = x2 2.7 10-6 = x2 1.6 10-3 = x2
3. PLUG into pH equations [OH-] = 1.6 10-3 M pOH = -log (1.6 10-3) pOH= 2.80 pH = 14.00 - 2.80 pH = 11.20
KaandKb Relationship Kaand Kb are related in this way: Ka Kb = Kw Therefore, if you know one of them, you can calculate the other.