Understanding the Lowest-Energy Configurations and Excited States in N2 Molecules
This text delves into the lowest-energy configurations of the N2 molecule, describing the first two excited states. It explains the complete occupation of electron shells, the characteristics of singlet and triplet states, and the configurations of two electrons in distinct molecular orbital (MO) shells. The discussion also includes how different arrangements can yield symmetric and antisymmetric spatial and spin components. Notably, it highlights the significance of the Franck-Condon factor in determining molecular states like ortho-H2 and para-H2.


Understanding the Lowest-Energy Configurations and Excited States in N2 Molecules
E N D
Presentation Transcript
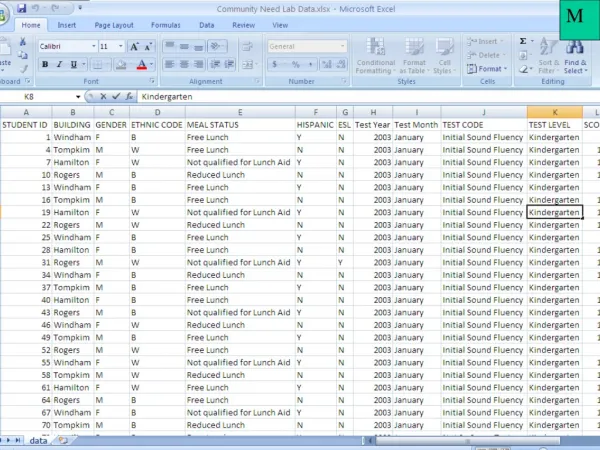
N2 Lowest-energy configuration First two excited configurations All shells fully occupied (N2): 1Sg+ (...) : Pg state (multiplicity, see later) (...) : is similar to hole states are like those for , i.e. (see table)
spin part spatial part Two electrons are located in 2 different MO shells: both singlet and triplet states exist Two electrons are located in same MO shell and same spatial orbital: only singlet state 3. Two electrons are located in same MO shell but different spatial orbitals: e.g. O2, most stable configuration L = l1+l2 = 0 Symmetric spatial part Antisymmetric spin part = singlet Antisym. spatial part Symmetric spin part = triplet
S+ or S- ? f1
e.g: ortho-H2 (I=1, Ns=3) para-H2 (I=0, Na=1)