Exploring Heat Transfer Experiments and Enthalpy Calculations
Investigate heat transfer through experiments, calculate enthalpy changes, and delve into Hess' Law, bond energies, and reactions. Learn concepts in chemical thermodynamics and calorimetry.

Exploring Heat Transfer Experiments and Enthalpy Calculations
E N D
Presentation Transcript
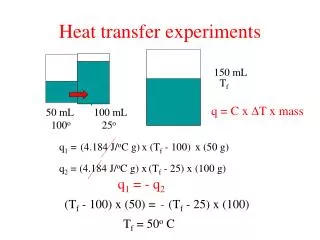
Heat transfer experiments 150 mL Tf q = C x T x mass 50 mL 100o 100 mL 25o q1 = (4.184 J/oC g) x (Tf - 100) x (50 g) q2 = (4.184 J/oC g) x (Tf - 25) x (100 g) q1 = - q2 (Tf - 100) x (50) = - (Tf - 25) x (100) Tf = 50o C
Enthalpy of reaction Hrxn= qrxn coffee cup calorimeter 10.5 g KBr in 125g water at 24o KBr(s) K+ (aq) + Br-(aq) Tf = 21o Calculate Hrxn qsystem = - qsurroundings = Hrxn
Hrxn qsystem = - qsurroundings = Hrxn T x mass qsurroundings = C x (4.184 J/goC) (21 - 24oC) (10.5 g + 125 g) qsurroundings = = -1756 J = +1756 J = Hrxn qsystem = - qsurroundings H is extensive a) endothermic b) exothermic Hrxn = 1756 J = 167 J/g = 19873 J/mol 10.5 g KBr
E = q + w E = q - PV At constant V, E = qv Bomb calorimeter qrxn = qsystem = -qcalorimeter T (oC) qcalorimeter = C (J / oC) x
Constant Volume calorimetry 2Fe (s) + 3/2 O2 (g) Fe2O3 (s) 11.2 g Fe(s), 1 atm O2 Ccalorimeter = 2.58 kJ/oC Tcalorimeter = + 31.9 oC -qcalorimeter qrxn = = Erxn = - (2.58 kJ/oC) (31.9oC) Erxn / 0.1 mol Fe2O3 = - 82.2 kJ = - 822 kJ/mol Fe2O3
Thermitereaction Al2O3(s) + 2Fe(l) 2Al(s) +Fe2O3(s) H is an Exothermic extensive, State function Hess’ Law
2Al(s) +Fe2O3(s) Al2O3(s) + 2Fe(l) 2Al(s) + 3/2 O2(g) Al2O3(s) H = -1676 kJ/mol _______________________________ __________ Fe2O3(s) 2Fe(s) + 3/2 O2(g) 2 Fe(s) + 3/2 O2(g) Fe2O3(s) H= - 822 kJ/mol + Al2O3(s) + 2Fe(s) -854 kJ/mol 2Al(s) + Fe2O3(s) 2 ( ) +15 kJ/mol 2 2 Fe(s) Fe(l) _______________________________ __________ 2Al(s) +Fe2O3(s) Al2O3(s) + 2Fe(l) Hrxn = -824 kJ/mol
Hess’ Law • Always end up with exactly the same reactants and products • If you reverse a reaction, reverse the sign of H • If you change the stoichiometry, change H
Heats of formation, Hof H = heat lost or gained by a reaction “o” = standard conditions: all solutes 1M all gases 1 atm “f” = formation reaction: 1mol product from elements in standard states for elements in standard states, Hof= 0
2Al(s) +Fe2O3(s) Al2O3(s) + 2Fe(l) products reactants elements Hof 2 Al(s) Al2O3(s) 2 Al(s) 2 Fe(s) Fe2O3 2 Fe (l) 3/2 O2(g) Hof Al2O3(s) + 2 HofFe (l) Al(s) Fe2O3
2Al(s) +Fe2O3(s) Al2O3(s) + 2Fe(l) products reactants elements Hof 2 Al(s) Al2O3(s) 2 Al(s) 2 Fe(s) Fe2O3 2 Fe (l) 3/2 O2(g) Hof Al2O3(s) + 2 HofFe (l) - Hof - Hof Fe2O3 Al(s) - Hrxn = nHofproducts nHofreactants
2Al(s) +Fe2O3(s) Al2O3(s) + 2Fe(l) - nHofreactants Hrxn = nHofproducts [HofAl2O3(s) + 2 HofFe(l)] Hrxn= - [HofFe2O3(s) + HofAl(s)] 2 [(-1676) + (15)] 2 - [(-822) + 0]kJ Hrxn = = -824 kJ
Bond Energies chemical reactions = bond breakage and bond formation bond energies positive energy required to break bond bond breakage a) endothermic b) exothermic (raise P.E.) bond formation exothermic (lower P.E.)
Bond energies CH4 (g) + 2O2 (g) CO2 (g) + 2H2O (g) Hrxn= bonds broken C-H 413 kJ O=O 495 kJ C=O 799 kJ O-H 467 kJ - bonds formed Hrxn= 4 [ (C-H) + (O=O)] 2 - [ (C=O) 2 + (O-H)] 4 = -824 kJ Hrxn= Hof products - Hof reactants =- 802 kJ
qv v.s. qp qp = H qv = E H = E + PV = E + nRT H = E + PV if n = 0 H = E 2Fe (s) + 3/2 O2 (g) Fe2O3 (s) n = (0 - 3/2) = - 3/2 H = (- 3/2)(8.314 x 10-3 kJ)(298) - 822 kJ/mol + H = -826 kJ/mol