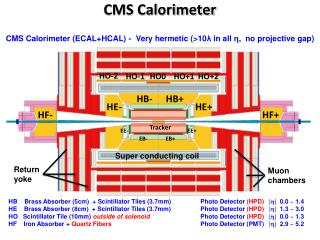
Determining ΔHrxn for the Reaction of Lead(II) Nitrate and Sodium Iodide in a Calorimeter
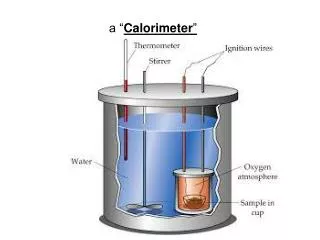
This experiment utilizes a coffee cup calorimeter to measure the heat exchanged during the reaction between lead(II) nitrate and sodium iodide, which produces lead(II) iodide and sodium nitrate. A 6.00-gram sample of sodium iodide is mixed with 200 mL of a 0.22 M lead(II) nitrate solution. Initially, the temperature of the solution is 22.6°C and rises to 24.2°C after the reaction. Using the specific heat and density of water for calculations, we will find the enthalpy change (ΔHrxn) for the reaction.

Determining ΔHrxn for the Reaction of Lead(II) Nitrate and Sodium Iodide in a Calorimeter
E N D
Presentation Transcript
A coffee-cup calorimeter is used to measure the heat exchanged when the following reaction is performed : Pb(NO3)2 + 2 NaI(aq) → PbI2(s) + 2 NaNO3(aq) Specifically… A 6.00-gram sample of sodium iodide is added to 200 mL of a 0.22 M lead(II) nitrate solution. Before the solid is added, the solution has a temperature of 22.6˚C. The temperature is recorded to be 24.2˚C when the reaction is complete. Use this information to determine the ΔHrxn. Assume that the specific heat and density of the mixture are the same as that for pure water.