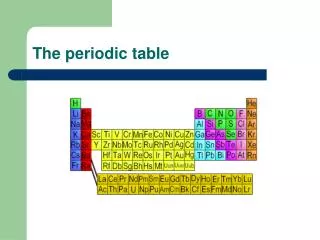
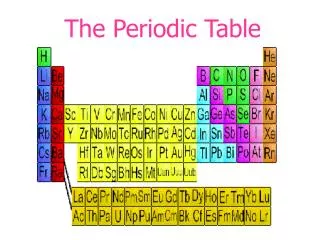
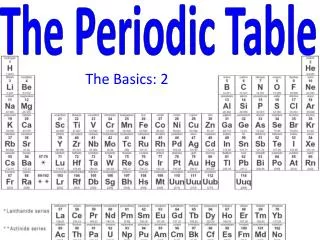
The Periodic Table
The Periodic Table. Biology Mr. Luis A. Velázquez. Dynamic Periodic Table. Element Symbols. All elements have a symbol . The symbol will always be a letter or two . Example: C, Ca. The symbol will always begin with a capital letter. .

The Periodic Table
E N D
Presentation Transcript
The Periodic Table Biology Mr. Luis A. Velázquez
Element Symbols • All elements have a symbol. • The symbol will always be a letter or two. Example:C,Ca • The symbol will always begin with a capital letter. • If the symbol has a second letter it will always • be lower case.
Atomic Number A unique number for each element that equals the number of protons in an atom of that element.
Atomic Mass Number A unique number for each element that equals the number of protons and neutrons present in the nucleus of the atom. In other words is the sum of the nucleus of the atom. (P+N=mass)
Practice 83 Bismuth Bi 209 209 What is the atomic mass? What is the atomic number? 83 83 What is the number of protons? What is the number of neutrons? 126
Elements A substance that cannot be broken down in to simpler substance. There 112elements in the periodic table.
Elements 112total elements 92 natural elements 20artificial or synthetic elements man-made elements.
Elements 25 needed for life 4 Are essential elements for life they make 99% of living tissue C, H , O, N 1 Element essential for life
State of Matter of Elements 1. Solid is a substance that has definite shape and definite volume. 2. Liquid is a substance that has no definite shape and has definite volume. 3. Gas is a substance that has no definite shape and no definite volume.
1. Some elements are found in nature as gases. 2. There are 11 elements as a gas state. 3.Gases can be classify as stable or unstable gases. Unstable gases: (5) gases H, N, O, F, Cl Stable gases: (6) gases He, Ne, Ar, Kr, Xe, Rn
Liquids 1. Some elements are found in nature as liquids. 2. There are 2 elements as liquid state. The two liquids are: Br, Hg
Summary There are 112elements in the periodic table, the are subdivide into: 1.Gases (11) H,N,O,Cl,F,He,Ne,Ar,Kr,Xe,Rn 2.Liquids (2) Br, Hg 3.Solids (99) Li, C,Mg,Mn,Co, Au,Ag,Cu,Fe…
Energy Potential and kinetic energy in elements: 1.Gases : have very high energy (KE) (PE) 2.Liquids : have regular energy (KE) (PE) 3.Solids: have very low energy (KE) (PE)
Answer the following Classify the following elements in Solid Liq or gas • ____He • ____C • ____Mn • ____Al • ____Cl • ____Br • ____H • ____Mg • ____Ca • ____N • ____Hg • ____Si • ____Xe • ____K Place an arrow to indicate the energy. Due Today…
Understanding the atom • The atomic number is the number of protons • The atomic mass number is the number of protons • and neutrons. The sum of the nucleus of the atom. 6 Atomic number = Protons = 6 C 12 Atomic mass = P + N = 12
Understanding the atom 17 Atomic number = Protons = 17 Cl Atomic mass = P + N = 35 35 17 P= N= 18 E= 17
Understanding the atom 11 Atomic number = Protons = 11 Na Atomic mass = P + N = 23 23 11 P= N= 12 E= 11
Understanding the atom 7 Atomic number = Protons = 7 N Atomic mass = P + N = 14 14 7 P= N= 7 E= 7
Understanding the atom 20 Atomic number = Protons = 20 Ca Atomic mass = P + N = 40 40 20 P= N= 20 E= 20
Answer the following Make a table to show the amount of protons, electrons, and neutrons for each of the following elements . • ____Ni • ____Zn • ____Cu • ____Al • ____Au • ____P • ____H • ____Mg • ____Ba • ____Rb • ____Hg • ____S • ____Xe • ____K 50 points
Classifing Elements 84 • Metals 17 • No Metal 8 • Metalloids At, Po, Te, Sb, As, Ge, Si, B
8A 1A 6A 7A 3A 4A 5A 2A No Reaction Transitional Metals Negative Negative Negative
Non- metals • Elements in nature that are found in the gas • stage are not metals. • There are 17 non- metals • He, Ne, Ar, Kr, Xe, Rn F, Cl Br, I Se, S, P, O, • N, C, H
Physical Properties of Metals • They show metallic luster. • They are opaque. • They have high density. • They are good conductors of heat and electricity. • They are solids (except mercury Hg) • They are malleable and ductile in their solid state.
Chemical Properties of Metals • They have one to four valence electrons. • They are good reducing agents. A reducing agent is an element that can add electrons to another element. • They form cations (positive ions) Example: Li+, Ca+2, Fe+3…
Physical Properties of Non-Metals • Poor conductors of heat and electricity. • Brittle - if a solid. • Nonductile. • Do not possess metallic luster. • Transparent as a thin sheet. • Solids, liquids or gases at room temperature.
Chemical Properties of Non-Metals • Usually have 4-8 electrons in their outer shell. • Gain or share valence electrons easily. • Form oxides that are acidic. • Are good oxidizing agents. • Have higher electronegativities.
The Valence Number • Column 1A has a valence number of 1. • Column 2A has a valence number of 2. • Column 3A has a valence number of 3. 1A 8A 3A 4A 5A 6A 7A 2A B series Transition Metals
Families of Elements • 1A Alkali Metals • 2A Alkaline Earth Metals • 3A Boron Family • 4A Carbon Family • 5A Nitrogen Family • 6A Oxygen Family • 7A Halogens Family • 8A Noble Gases
Alkali Metal Family • Column 1A ( Except Hydrogen) • Li • Na • Cs • Rb • Fr • They are the most reactive elements in the periodic • table.
Naming Compounds • Metals are always positive. • Metals are always written first. • Metals are name the same way as the element. • Non-Metals are always negative. • Non-Metals are always written second. • The names of non-metals are always ending in ide .
Naming Compounds • Example: CaO • Metals = Calcium (Ca) • Metals are name the same way as the element. • Non-Metals = Oxygen (O). • The names of non-metals are always ending in ide . • The names of non-metals oxide . • The names of this compound is: Calcium Oxide
Naming Compounds • Example: NaCl • Metals = Sodium (Na) • Metals are name the same way as the element. • Non-Metals = Chlorine (Cl). • The names of non-metals are always ending in ide . • The names of non-metals chloride . • The names of this compound is: Sodium Chloride
Naming Compounds • Example: MgF2 • Metals = Magnesium (Mg) • Metals are name the same way as the element. • Non-Metals = Fluorine (F). • The names of non-metals are always ending in ide . • The names of non-metals Fluoride . • The names of this compound is: MagnesiumFluoride
Naming Non-metals • Fluorine = Fluoride • Chlorine = Chloride • Bromine = Bromide • Iodine = Iodide • Oxygen = Oxide • Sulfur = Sulfide • Phosphorous = Phosphide
Naming Compounds Problems Write the correct names for the following compounds . BaCl2 NaF Ag2O CuBr FeO MgS Al2O3 CaI2 BeS Hg2I2 CrCI3 K2S 60 points