The Periodic Table
Explore the historical development and modern interpretation of the periodic table through key scientists and concepts. Understand periodic trends, elements' classifications, and the significance of atomic properties.

The Periodic Table
E N D
Presentation Transcript
History of the Periodic Table: • Prior to 1650, there were 11 known elements: • Ag, As, Au, C, Cu, Fe, Hg, Pb, S, Sb, Sn • By 1800, about 30 elements were known • By 1850, about 60 elements were known • By 1900, about 80 elements were known • By 1950, about 90 elements were known • Now, over 100 elements are known
The Scientists: • John Newlands (1864) • Arranged elements by atomic mass • Saw that every 8th element had similar properties • Law of “octaves” • If chemical elements are arranged according to increasing atomic mass, those with similar physical and chemical properties occur after each interval of seven elements.
The Scientists: • Dmitri Ivanovich Mendeleev (Russian Chemist) • 1st recognizable periodic table in 1869 • Ordered by atomic mass • Grouped elements by physical and chemical properties. • Predicted new elements
The Scientists: • Sir William Ramsay • Discovered Ne, Ar, Kr, Xe around 1900 (noble gases)
The Scientists: • Henry Mosely (British Physicist) • Determined the number of protons in each element (1913) • Rearranged the periodic table by atomic number • Better than using atomic masses of elements.
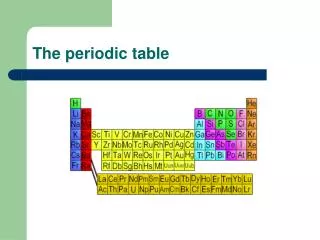
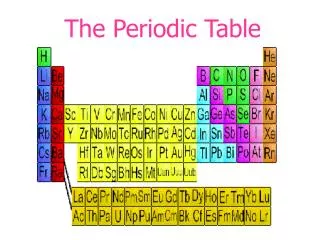
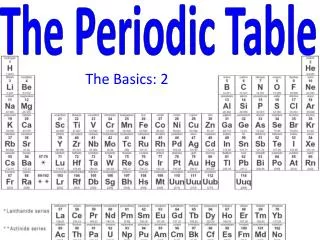
The Periodic Table Today: • Arranged in order of increasing atomic number. • 7 rows called “periods” • 18 columns called “groups” or “families” • Also Lanthanides and Actinides • Representative Elements (main group elements) • s-block and p-block • Transition Metals • d-block • Inner Transition Metals • f- block
The Periodic Table Today: • Noble Gases • Farthest right column • All gases • Very unreactive (used to be called “inert gases”)
The Periodic Table Today: • Nonmetals • Right of staircase • Poor conductors of heat and electricity • Not very lustrous (shiny) • Solids are brittle
The Periodic Table Today: • Metalloids/semi-metals • On the staircase • B, Si, Ge, As, Sb, Te, Po • Have both metal and nonmetal properties • Moderate conductivity (used in semiconductors)
The Periodic Table Today: • Metals • All the rest (leftof the staircase) • Conductors of heat and electricity • Lustrous (shiny) • Ductile (can be drawn into a wire) • Malleable (can be hammered into any shape)
Periodic Law: • Properties of elements change in a periodic fashion with increasing atomic number. • Every period the properties start over • Properties will repeat • Elements in the same group have similar properties! • Why? • Valence electrons!
Periodic Law: • Octet Rule – atoms tend to gain or lose electrons until they have 8 valence electrons (an “octet”). • Noble gases already have 8 valence electrons. • Stable octet = • 8 electrons in the outer PEL • Very Stable
Obtaining a Stable Octet: • Ion = an atom with a positive or negative charge • Formed by atoms gaining or losing electrons. • Never formed by gaining or losing protons • Cation = positive ion • Anion = negative ion
Periodic Trends: • Atomic Radius (A.R.) = “size” of the atom
Periodic Trends: • Ionization Energy (I.E.) = amount of energy that is required to remove an electron from an atom. • Results in a cation • Cations are smaller than their parent atom, because you are removing an electron.
Periodic Trends: • Electron Affinity (E.A.) = how much an atom “wants” an electron. • Results in anions • Anions are larger than their parent atom (adding an electron)
Which element has the Largest A.R.? Which has the smallest A.R.? Smallest A.R. Largest A.R.
Which element has the lowest I.E.? Which has the highest I.E.? Highest I.E. Lowest I.E
Which element has the lowest E.A.? Which has the highest E.A.? Highest E.A. Lowest E.A.