Understanding the Periodic Table: Organizing Elements and Their Properties
This comprehensive overview of the Periodic Table explains the organization of elements into three broad classes: metals, nonmetals, and metalloids. It delves into the concept of an element as a fundamental substance made of atoms, detailing atomic structure involving protons, neutrons, and electrons. You'll learn about atomic numbers, isotopes, and the significance of Mendeleev's contributions to the table's current arrangement. Discover how elements and their properties are systematically organized, aiding scientists and chemists in understanding chemical behaviors and interactions.


Understanding the Periodic Table: Organizing Elements and Their Properties
E N D
Presentation Transcript
Organizing the Elements • OBJECTIVES: • Identify three broad classes of elements.
Organizing the Elements • OBJECTIVES: • Explain how elements are organized in a periodic table.
What is an Element? • Before we begin let us first talk about what an element is… • An element is a material which cannot be broken down or changed into another substance using chemical means • they are made of atoms • They are floating all around us all the time • They are what makes life function • Without elements you would not exist
Remember the Atom • Atom- • The basic unit of matter made up of three subatomic particles • Protons- positively charged • Located in the nucleus • Neutrons- no charge • Located in the nucleus • Electrons- negatively charge • Encircling the nucleus
The Atom • Now the protons and neutrons stay in the nucleus and most often do not undergo reactions • When the nucleus is disrupted the results are severe… • i.e. Star Formation Nuclear explsion
The Atom • That leaves us with the electron… • Electrons float around the nucleus • They reside in levels and the outermost level is the valance level/shell • This is where the valance electrons reside • These are the electrons in the outermost shell and these are the electrons that undergo chemical reactions • Now remember these electrons can be lost or gained resulting in a change in charge of the atom (more on this later)
Elements • So back to the elements for a moment • Elements are represented by symbols • The elemental symbols are either a single letter or a series of letters (2 or 3) • A capital letter always represents a new element • If an element has another letter the second/third letters are always lower case • i.e. • C represents Carbon • Co represents Cobalt • CO represents Carbon and Oxygen (Carbon Monoxide)
Atomic Number? • So what is the Atomic Number? • The atomic number is the atom’s number • It is the number of protons in a given element • Therefore the number of protons always defines the element • For neutral atoms the atomic number is also the number of neutrons and electrons • Because neutral atoms will have the same number of electrons and protons • And unless the atom is an isotope the number of protons and neutrons are always the same
Useful Definitions • Isotope- an atom with a different number of neutrons than protons • Neutrons have been lost or gained • Mass Number- the mass of an atom • made up of the neutrons and protons • The Periodic Table of Elements actually use the average mass of all the isotopes of the element
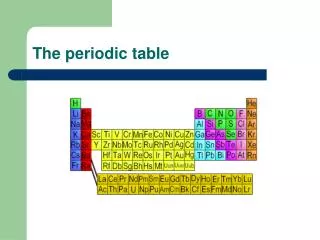
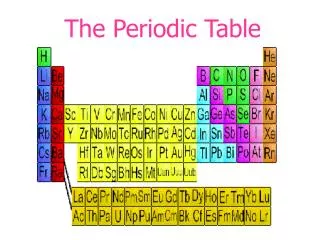
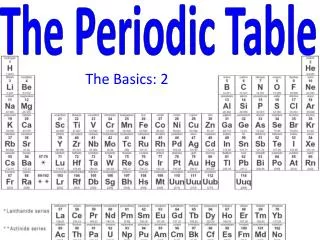
Back to the Periodic Table The elements are all arranged for us on the Periodic Table of Elements
Why is the Periodic Table important to me? • The periodic table is the most useful tool to a chemist. • You get to use it on every test. • It organizes lots of information about all the known elements.
Pre-Periodic Table Chemistry … • …was a mess!!! • No organization of elements. • Imagine going to a grocery store with no organization!! • Difficult to find information. • Chemistry didn’t make sense.
HOW HIS WORKED… Put elements in rows by increasing atomic weight. Put elements in columns by the way they reacted. SOME PROBLEMS… He left blank spaces for what he said were undiscovered elements. (Turned out he was right!) He broke the pattern of increasing atomic weight to keep similar reacting elements together. Dmitri Mendeleev: Father of the Table
The Current Periodic Table • Mendeleev wasn’t too far off. • Now the elements are put in rows by increasing ATOMIC NUMBER!! • The horizontal rows are called periods and are labeled from 1 to 7. • The vertical columns are called groups are labeled from 1 to 18.
Organizing the Elements • A few elements, such as gold and copper, have been known for thousands of years - since ancient times • Yet, only about 13 had been identified by the year 1700. • As more were discovered, chemists realized they needed a way to organize the elements.
The Periodic Law says: • When elements are arranged in order of increasing atomic number, there is a periodic repetition of their physical and chemical properties. • Horizontal rows = periods • There are 7 periods • Vertical column = group (or family) • Similar physical & chemical prop. • Identified by number & letter (IA, IIA)
Areas of the periodic table • Three classes of elements are: 1) metals, 2) nonmetals, and 3) metalloids • Metals: electrical conductors, have luster (shine), ductile, malleable • Nonmetals: generally brittle and non-lustrous, poor conductors of heat and electricity
Areas of the periodic table • Properties of non-metals • Some nonmetals are gases (O, N, Cl) • some are brittle solids (S) • one is a fuming dark red liquid (Br) • Notice the heavy, stair-step line? • Metalloids/Semimetals: border the line-2 sides • Properties are intermediate between metals and nonmetals
Classifying the Elements • OBJECTIVES: • Describe the information in a periodic table.
Squares in the Periodic Table • The periodic table displays the symbols and names of the elements, along with information about the structure of their atoms: • Atomic number and atomic mass
What does the information in the box tell me? 1 H Hydrogen 1.008 Atomic Number = # of protons Elemental Symbol Atomic Mass = # of protons plus neutrons Element Name
Groups of elements - family names • Group IA – alkali metals • Forms a “base” (or alkali) when reacting with water (not just dissolved!) • Group 2A – alkaline earth metals • Also form bases with water; do not dissolve well, hence “earth metals” • Group 7A – halogens • Means “salt-forming”
The Groups of the Periodic Table • Group 1: The Alkali Metals • Most reactive metals on the PT • Rarely found free in nature • Charge of 1—1 valence electron • Group 2: The Alkaline Earth Metals • Still quite reactive • Charge of 2—2 valence electrons
T he Groups of the Periodic Table • Groups 3-12: Transition Metals • Found freely and in compounds in nature • Charge is usually 2 but can vary—usually 2 valence electrons • Group 13: Boron Family • Charge is 3—3 valence electrons
The Groups of the Periodic Table • Group 14: The Carbon Family • Contains elements that can form unusual bonds (carbon and silicon) • Charge is +4 or -4—contains 4 valence electrons • Group 15: The Nitrogen Family • Charge is -3—contains 5 valence electrons
The Groups of the Periodic Table • Group 16: The Oxygen Family • Also known as the chalcogens • Charge is -2—6 valence electrons • Group 17: The Halogens • Most reactive nonmetals • charge is -1—7 valence electrons • Group 18: The Noble Gases (The Inert Gases) • Nonreactive • Charge is 0—2 or 8 valence electrons
The Periodic Table Column = Group or Family 18 columns on the Periodic Table Row = Period 7 rows on the Periodic Table
Metals, Nonmetals, and Semi-metals Only nonmetal on the metal side Nonmetals are on the right of the stair-step Metals are to the left of the stair- step Semi-metals, “metalloids,” touch the stair-step
Special Rows on the PT Lanthanides Actinides
Classifying the Elements • OBJECTIVES: • Distinguish representative elements and transition metals.
Groups…Here’s Where the Periodic Table Gets Useful!! • Elements in the same group have similar chemical and physical properties!! • (Mendeleev did that on purpose.) • Why?? • They have the same number of valence electrons. • They will form the same kinds of ions.
Families on the Periodic Table • Columns are also grouped into families. • Families may be one column, or several columns put together. • Families have names rather than numbers. (Just like your family has a common last name.)
8A 1A 2A 3A 4A 5A 6A 7A • Elements in the 1A-7A groups are called the representative elements outer s or p filling
These are called the inner transition elements, and they belong here The group B are called the transition elements
Hydrogen • Hydrogen belongs to a family of its own. • Hydrogen is a diatomic, reactive gas. • Hydrogen was involved in the explosion of the Hindenberg. • Hydrogen is promising as an alternative fuel source for automobiles
Alkali Metals • 1st column on the periodic table (Group 1) not including hydrogen. • Very reactive metals, always combined with something else in nature (like in salt). • Soft enough to cut with a butter knife
H Group 1A are the alkali metals (but NOT H) Group 2A are the alkaline earth metals
Alkaline Earth Metals • Second column on the periodic table. (Group 2) • Reactive metals that are always combined with nonmetals in nature. • Several of these elements are important mineral nutrients (such as Mg and Ca
Transition Metals • Elements in groups 3-12 • Less reactive harder metals • Includes metals used in jewelry and construction. • Metals used “as metal.”
Boron Family • Elements in group 13 • Aluminum metal was once rare and expensive, not a “disposable metal.”
Carbon Family • Elements in group 14 • Contains elements important to life and computers. • Carbon is the basis for an entire branch of chemistry. • Silicon and Germanium are important semiconductors.
Nitrogen Family • Elements in group 15 • Nitrogen makes up over ¾ of the atmosphere. • Nitrogen and phosphorus are both important in living things. • Most of the world’s nitrogen is not available to living things. • The red stuff on the tip of matches is phosphorus.